Often used to ameliorate pain, non-steroidal anti-inflammatory drugs (NSAIDs) are also routinely recommended by doctors, therapists and Dr Google to “reduce the inflammation” when you incur a musculoskeletal strain or injury.
The oft-cited rationale for quelling inflammation with NSAIDs is “so the body can begin the healing process.”
Today, you’re going to learn why the research does not support this contention. Further, you’ll learn why this junk is more likely to send you to hospital than exert any positive impact on your physique or performance.
How can that be?
Well, let’s start with the entire ‘anti-inflammatory’ premise.
Inflammation is often spoken of as if it were a disease.
It is not.
Inflammation is part of the body’s response to injury or disease.
Nature didn’t evolve the inflammatory process for shits and giggles. Inflammation is an important part of the healing process.
Anti-Inflammatory, Anti-Healing?
To understand why NSAIDs might be detrimental to muscle, connective tissue and bone healing, we need to understand their mechanisms of action.
Let’s start with their effects on muscle. This section involves fancy, sciencey-sounding words, but I swear upon a tray of Sicilian cannoli it will be brief.
NSAIDs inhibit cyclooxygenase (COX) enzymes, which are responsible for the production of prostaglandins - a group of lipids with hormone-like actions. Your body produces prostaglandins primarily at sites of tissue damage or infection (Bjorkman, 1998; Funk, 2001).
Prostaglandins are a key part of the inflammatory process, one of their functions being to instigate the activity of satellite cells (Brewer & Waddell, 2012).
Satellite cells are specialized stem cells located in skeletal muscle that play a crucial role in muscle repair and regeneration. Satellite cells remain dormant until activated after injury, at which point they proliferate and differentiate to form myoblasts - another class of stem cells that then fuse and form multinucleated muscle cells known as myotubes, which in turn mature into functional muscle fibers.
In summary, COX > prostaglandins > satellite cells > myoblasts > myotubes > functional muscle fibers > back to normal activity > happy days.
So anything hampering the function of prostaglandins and satellite cells might be best avoided by those seeking optimal injury repair and muscle growth.
That’s the sciencey-sounding bit, I told you it would be brief. Now give me my cannoli.
NSAIDs vs COX and Satellite Cells
The earliest human study I’m aware of looking at this issue in a controlled manner was Reynolds et al (1995). Forty-four patients with acute hamstring tears were randomly allocated to one of three treatment groups: meclofenamate (100 mg x 3 times a day), diclofenac (50 mg x 3 times a day) and placebo.
All patients received the same intensive physiotherapy treatment over the 7-day treatment period. Patient assessments were performed on days 1, 3 and 7 of the 7-day study period and included pain assessment, swelling measurement and isokinetic muscle performance testing.
So what happened?
All three groups experienced similar improvement over the seven days. There was no difference in swelling between groups. When measured against the non-affected leg, ratios for peak torque, average power and total work during maximal isokinetic exercise did not differ between groups.
There was no significant difference in pain scores between groups, a curious finding considering a major selling point of NSAIDs is pain alleviation. In fact, the pain scores for the placebo group tended to be lower on days 3 and 7, but the difference did not attain statistical significance. When only patients with severe injuries were included in the analysis, the difference in pain scores at day 7 became statistically significant. Subjects with severe hamstring injuries receiving placebo demonstrated a steeper reduction in pain at day 7 than did subjects in either of the NSAID groups.
Adverse events, mainly gastrointestinal, were experienced by 37% of the NSAID subjects; 14% of placebo subjects reported adverse events (drowsiness and increased stool frequency).
The researchers concluded NSAID administration may “have delayed the pain recovery in those with the most severe injuries.”
“Accordingly, we suggest that in view of the cost, adverse effects and risks of delayed healing, NSAIDs should not be prescribed routinely for acute muscle injuries until such time as convincing evidence for their efficacy in this condition becomes available.”
This plea was largely ignored.
Subsequent Human Research
Short-term research by Mackey et al (2007) and Mikkelsen et al (2009) found evidence of reduced satellite cell activity after indomethacin administration in volunteers that performed heavy eccentric exercise or a long-distance run. However, Paulsen et al (2010) found no difference in satellite cell activity differences between the celecoxib and placebo groups after performance of maximal eccentric biceps curls.
Researchers have also examined the acute effects of NSAIDs of post-training muscle protein synthesis, the process in which new muscle proteins are produced . After a bout of heavy resistance exercise, the muscles you trained will experience an increase in protein synthesis (Chesley et al, 1992; Phillips et al, 1997). If you’re eating enough protein and calories and getting enough rest, then muscle protein synthesis should outweigh muscle protein breakdown, the net effect being that your muscles get bigger and stronger.
Trappe et al (2002) observed the expected increase in muscle protein synthesis after heavy eccentric exercise in young males taking a placebo. However, it was severely blunted in those receiving either the maximal OTC dose of ibuprofen (1,200 mg/day) or acetaminophen (4,000 mg/day).Post-exercise levels of prostaglandin F2 alpha levels also significantly increased in the placebo group, but not in the the ibuprofen and acetaminophen groups (Trappe et al, 2001) .
Peterson et al (2011a) also found lower post-exercise prostaglandin F2α levels with ibuprofen compared with placebo, but no difference in muscle or tendon collagen protein synthesis.
Burd etal (2010), meanwhile, found no difference between celecoxib or placebo in muscle protein synthesis after a bout of leg extensions.
Conflicting results like these are frustrating and not exactly helpful when trying to get a clear answer to a pressing question. So let’s forget about short-term studies looking at satellite cells, prostaglandins and protein synthesis, and see whether anyone has actually studied the longer-term effects of these drugs on strength and hypertrophy.
NSAIDs versus the Noble Quest to Get Jacked
Incredibly, no-one has yet published a long-term study examining the effect of NSAIDs on the kind of resistance training people perform in real life – you know, multiple upper and lower body exercises with a focus on compound movements.
Considering how long NSAIDs have been available and freely administered to athletes, this is a disgrace.
All we presently have are a handful of small studies that examined the effect of NSAIDs on subjects performing single exercises (usually leg extensions or biceps curls) for up to 12 weeks. Even this less-than-optimal research took a bloody long time to begin showing up in the journals.
Krentz et al (2008) performed a study with males and females in their 20s, in which their right and left biceps were trained on alternate days for 6 weeks. In a double-blind design, they were randomized to receive 400 mg of ibuprofen immediately after training their left or right arm, and a placebo after training the opposite arm the following day. Both arms evinced increased muscle thickness, 1RM strength and soreness, with no difference between placebo and ibuprofen.
Petersen et al (2011b), using older patients with knee osteoarthritis, compared the effects of ibuprofen, glucosamine or placebo during 12 weeks of quadriceps-only strength training. The subjects performed unilateral leg extensions and leg presses 3 times weekly.
No differences between groups were observed in gains in muscle cross-sectional area. Compared to placebo, ibuprofen produced small increases in maximal isometric strength and maximal eccentric muscle strength but no difference in 5-RM leg press or 5-RM knee extension.
Trappe et al (2011), using folks in their mid-late 60s, found OTC doses of acetaminophen or ibuprofen, in combination with resistance training, did not inhibit and in fact appeared to enhance muscle hypertrophy and strength gains in older adults compared to placebo.
The study involved performance of only leg extensions three times per week, over a 12-week period. The placebo subjects were a mean three years older and 5-13 kg lighter than the drug group subjects, although it’s unclear if and how this affected the results.
Mallinson et al (2025) randomized seventeen trained males (mean 24.5 years) to consume either placebo or diclofenac (75 mg) daily for 12 weeks and perform 3 × 30 maximal, isokinetic knee extensions in the non-dominant leg thrice each week. The NSAID group experienced greater increase in quadriceps muscle cross sectional area at 84 days (placebo 3.9%, NSAID 8.6%), but this was not accompanied by greater strength gains.
A curious finding of this study was that muscle CSA stagnated from day 28 to 84 in the placebo group (4.3% versus 3.9%, respectively), yet almost doubled during the same time frame in the diclofenac group (4.6% vs 8.6%, respectively).
To claim that, in a real life scenario involving a whole-body routine, your muscle gains would stall at 28 days but proceed to double through day 84 on the same routine simply by virtue of taking an NSAID, is nonsensical. To start making gains on a non-productive routine means you either weren’t eating enough protein and/or calories and finally got your dietary sh*t together, or you commenced anabolic steroid use.
Let’s face it, no-one in their right mind would seriously suggest you can make steroid-like gains simply by taking NSAIDs. Well, dipshit influencers might (more on that shortly), but dipshit influencers can hardly be considered a trustworthy source of drug and training information.
Contrary to these results, Lilja et al (2018) found a maximal ibuprofen dose impaired strength and hypertrophy in healthy men and women aged 18-35 years. The subjects were randomly assigned to daily consumption of ibuprofen (1200 mg) or aspirin (75 mg) for 8 weeks. During this time, subjects completed supervised knee-extensor resistance training in which one leg was worked with a flywheel ergometer, the other with weight-stack training. The increase in quadriceps volume was similar between the two training modalities, but greater with aspirin (7.5%) compared with ibuprofen (3.7%). In the weight stack-trained legs, muscle strength improved similarly, but in the flywheel legs, increases (10-23%) in muscle strength were generally greater for aspirin. The problem with this study was there was no placebo group and both intervention groups took an NSAID.
And that’s the sum total of the trial research looking at the longer-term effects of NSAIDs on strength and hypertrophy: None involved a ‘real world’ training routine involving multiple body parts; a couple found improvements of questionable veracity and doubtful clinical significance; and overall the findings indicate NSAIDs do a whole lot of nothing to improve your muscle and strength gains.
But They Alleviate Soreness, Right?
Again, the findings are mixed. A 2024 study published in the Journal of Science and Medicine in Sport found NSAIDs do not prevent exercise-induced performance deficits or alleviate muscle soreness (Roberts et al, 2024). The randomized, double-blind, crossover study examined the effect of consuming a single dose of each of three NSAIDs (celecoxib, 200 mg; ibuprofen, 800 mg; flurbiprofen, 100 mg) or placebo 2 hours before an a plyometric training session. Twelve healthy adults, aged 18–42 years, took part.
At 4-hours and 24-hours post-exercise, there were no significant differences in total work, heart rate, rating of perceived exertion, muscle soreness or vertical jump performance between treatments. Additionally, no significant differences in were observed between treatments. The only remotely positive finding was that celecoxib attenuated decreases in maximum voluntary contraction force at 4-hours post exercise. That’s a finding of doubtful benefit considering the limited and specific time frame – few sensible people would be attempting to exert maximal force a mere four hours after a plyo workout.
Küster et al (2013) found consumption of NSAIDs before a marathon was associated with increased joint and muscle pain after the race.
The Undeserved Popularity of NSAIDs
Like acetaminophen, NSAIDs are readily available without a prescription in supermarkets and pharmacies. Sitting in a grocery store alongside dental floss and bath salts imparts NSAIDs with a disarming aura of safety.
As a result of their widespread availability and perceived safety (in the eyes of layfolk), NSAID use is becoming increasingly pervasive.
According to ClinCalc, over 58 million prescriptions were written for NSAIDs in the US in 2023 alone. The annual number of prescriptions steadily declined from 2014, while the number of patients receiving NSAID scripts has remained relatively stable. This would suggest doctors are prescribing lower amounts of NSAIDs per patient – perhaps a lingering effect of the Vioxx and Bextra scandals of the mid-noughties.
These prompted the pharma-friendly FDA to reluctantly “ask” NSAID manufacturers “to include a boxed warning, highlighting the potential for increased risk of cardiovascular (CV) events and the well described, serious, potential life-threatening gastrointestinal (GI) bleeding associated with their use.”
So the Vioxx and Bextra scandals and subsequent label warnings appear to have tempered doctors’ NSAID prescribing habits. But while US doctors are prescribing lower amounts of NSAIDs, they are increasingly prescribing other problematic drugs for pain treatment, including acetaminophen, antidepressants and gabapentinoids (Gorfinkel et al, 2022).
Joe and Jane Public, meanwhile, never got the memo. More people than ever are consuming NSAIDs. Obviously, if doctors are prescribing lower NSAID amounts, it means increased consumption of these drugs is a result of non-prescription over-the-counter purchases.
A 2013 survey of 1,750 US adults conducted by Harris Interactive found NSAIDs were used by 63% of respondents in the previous 12 months. Extrapolated to the US population for 2013, that equated to 200 million American adults using NSAIDs annually. The authors noted actual use of NSAIDs in this population may have been higher because 15% of respondents were unsure if they used NSAIDs.
The authors compared their results with a 1997 survey that found only 17% of respondents used NSAIDs in the previous 12 months. As the percentage of prescription users remained constant since the 1997 survey, in line with the ClinCalc data, the dramatic increase since 1997 stemmed from OTC users.
A 2022-2023 survey of over 13,000 Australian households found 20.6% used ibuprofen in the last two weeks. That same survey, by the way, found 43.5% had used paracetamol in the same time frame.
Interestingly, 13.4% of those who used ibuprofen reported no bodily pain during the four weeks prior to interview, while 22.6% reported “very mild/mild” pain.
Which begs the question as to why these people were even taking NSAIDs in the first instance?
Part of the explanation is that, like most Anglo-dominant countries, Australia has long possessed a reckless attitude towards ingesting drugs; witness the country’s idiotic drinking culture and its ranking alongside New Zealand and North America as the world’s most prolific user of illicit recreational drugs (fun facts: Australia is now the world’s biggest per capita consumer of cocaine, and along with the UAE and Saudi Arabia, has the world’s highest ‘retail’ cocaine prices).
The other likely reason Australians take NSAIDs even when experiencing zero pain is the pervasive but false perception that NSAIDs (and pain relievers like paracetamol) are relatively harmless, innocuous drugs.
The bottom line is that increasing numbers of people are using NSAIDs, and most appear to be self-prescribing.
More Dangerous than You Think
Most NSAIDs available today are what as known as non-selective COX inibitors, meaning they inhibit both COX-1 and COX-2 enzymes. Drugs like aspirin, indomethacin, and meclofenamate all fall into this class, although they tend to be more COX-1 specific. The popular ibuprofen, meanwhile, is a ‘neutral’ non-selective COX inhibitor.
In response to the adverse effects of these drugs, such as bleeding and gastrointestinal issues, Big Pharma reached into its lucrative “new and improved” hat and pulled out a class of drugs known as COX-2 inhibitors. These new NSAIDs were aggressively marketed as a whizz-bang-woo-hoo advancement on their non-selective predecessors, but quickly proved to be anything but.
Examples of COX-2 inhibitors include Vioxx (rofecoxib) and Bextra(valdecoxib), which were removed from the market in 2004 and 2005, respectively, because they were very effective – at causing heart attack and stroke.
Lumiracoxib was never approved by the FDA, but was OK’d by numerous other regulatory agencies around the world, including Australia’s hopelessly corrupt and dishonest Therapeutic Goods Administration (TGA). In 2007, the pharma-funded TGA cancelled the registration of lumiracoxib due to concerns it may cause liver failure. Canada and several EU countries followed suit shortly afterwards.
Lumiracoxib was manufactured by Novartis and is still sold in a few countries, including Mexico, Ecuador and the Dominican Republic, under the trade name Prexige.
Celecoxib (available as Celebrex and generic formulations) now remains the only COX-2 inhibitor available in the US. Other COX-2 inhibitors available outside the US include meloxicam, etoricoxib, parecoxib and the aforementioned lumiracoxib.
Adverse Drug Reactions, Anyone?
Like the liver-toxic acetaminophen, NSAIDs are a leading cause of adverse drug reaction (ADR) events and should be used with great caution (if at all).
NSAIDs increase the risk of acute and long-term kidney damage (Lim et al, 2025).
From the first day of use, all NSAIDs increase the risk of gastrointestinal (GI) bleeding, myocardial infarction, and stroke. All NSAIDs increase both bleeding and cardiovascular disease risk; selective COX-2 inhibitors are more likely to cause cardiovascular events, whereas less selective NSAIDs are more likely to cause GI bleeds. The risk of bleeding and CVD events is considerably higher in older people, many of whom take medicines known to interact with NSAIDs (Davis & Robson, 2016).
Imagine ending up in coronary care after popping NSAIDs because your elbow was a bit sore after a triceps workout. Or, in the case of Australia, because your elbow wasn’t sore at all. On a risk:reward scale of 1-10, that would rate “WTF was I thinking?!” and lead to a protracted bout of self-kicking.
As if all that isn’t bad enough, NSAIDs have been linked to erectile dysfunction, although the research is mixed (Li et al, 2018). There is, however, at least one case report that leaves little doubt NSAIDs can negatively affect at least some men in this regard. A 2008 report documented the case of an otherwise healthy 28 year old male who took celecoxib for periodic dental pain. The man presented for medical attention after two weeks of his dinger refusing to dong; he had been sexually active with his wife for three years prior. After ceasing use of celecoxib, he resumed his potency and regular intercourse. To confirm if celecoxib was the causal agent, the doctors asked him to take the same drug if his dental pain occurred again and report to them if the erectile dysfunction reappeared. After 2 months, the pain re-occurred and he took celecoxib – and again experienced erectile difficulties (Dhikav et al, 2008).
While indisputably a problematic class of drug, there is evidence to suggest NSAID adverse events severe enough to require hospitalization are declining in some countries. However, NSAID use still remains one of the leading causes of ADR-related hospital admissions.
A systematic review published in 2008 implicated NSAIDs in 14.6% and 18.8% of ADR-related hospital admissions for younger and older adults, respectively (Kongkaew et al, 2008).
A more recent 2023 systematic review found NSAIDs accounted for 6.2% of ADR-related hospital admissions (Haerdtlein et al, 2023).
It may be a different story for youngsters. Big Pharma, always looking to expand its markets, has succeeded in getting more children and adolescents on NSAIDs. In Italy, the proportion of pediatric ibuprofen packages purchased without a prescription rose from 28% in 2008 to 70% in 2015. From 2020 to 2024, pediatric ibuprofen use grew by over 60% and, not surprisingly, the number of reported ADRs also increased substantially (Vassallo et al, 2025).
Athletes Face Extra Risks
Clearly, Bob and Barbara Average are at risk of a range of NSAID side effects. Unfortunately, athletes face a few additional complications.
Gut Busters
NSAID use can cause gastrointestinal distress, something most endurance athletes prefer to avoid during an important event. A 2012 study published in Medicine & Science in Sports & Exercise found ibuprofen aggravated exercise-induced small intestinal injury and induced gut barrier dysfunction in healthy, trained men performing stationary cycling (Van Wijck et al, 2012).
As part of the 2010 Bonn marathon, participants were invited to complete questionnaires about their training and racing history, medication use, adverse events and premature withdrawal during the race. The response rate was quite good; 3,913 of 7,048 participants returned their questionnaires (Küster et al, 2013).
Nearly half of the study cohort used analgesics before the actual race (n = 1,931, 49%) while the remainder reported not having used any. The most frequently used analgesic was diclofenac, used by 47% of the analgesics cohort before the race; 11% resorted to supra-OTC doses (over 100 mg). The second most commonly used analgesic was ibuprofen, and 43% of those who took ibuprofen ingested ≥800 mg (twice the recommended OTC single dose). Aspirin was used less frequently, mostly at low therapeutic doses.
No significant difference was found between the analgesics cohort and controls in terms of premature race withdrawal overall, but intestinal cramps were significantly more frequently blamed for race withdrawal in the analgesics cohort.
Joint and muscle pain after the race were significantly more frequent in the analgesics cohort.
Disturbingly, the analgesics cohort displayed a 5-fold higher risk of adverse events. This elevated risk rose significantly with increasing analgesic dose.
Nine respondents reported temporary hospital admittance: three for temporary kidney failure (post-ibuprofen ingestion), four with bleeds (post-aspirin ingestion) and two cardiac infarctions (post-aspirin ingestion).
None of the analgesic-free respondents reported hospital admittance.
Bone Weakeners
Researchers have observed a strong correlation between regular ibuprofen/naproxen/celecoxib use and stress fracture incidence in the general population (Ciuciu et al, 2024). Given that athletes in impact sports like running are at increased risk of stress fractures, this is an additional cause for concern.
By the way, if you’re recovering from a bone fracture or break, be aware that reviews and meta-analyses on the issue have found “the literature indicates a negative effect of NSAIDs on bone healing” (Wheatley et al, 2019) and “an adverse effect on bone tissue” (García-Martínez et al, 2015).
Kidney Krushers
Then there’s the detrimental effect of NSAIDs on kidneys. Long-distance athletes who train and/or compete in the heat are already at risk of kidney damage because of dehydration.
A multi-site, randomised, double-blind trial compared participants in 50-mile (80 km) foot races in the Gobi, Atacama, Ecuador and Sri Lankan deserts to determine the impact of NSAID use (Lipman et al, 2017). Participants were randomised to either ibuprofen (400 mg) or placebo taken every 4 hours after the start of the fifth stage, and were offered equal amounts of water during the race. The average amount of ibuprofen ingested was 1200 mg, with 75% of participants taking between 1200 and 1800 mg.
Runners who ingested ibuprofen had a greater absolute rate of acute kidney injury, and the severity of injury was also worse in the ibuprofen group than the placebo group.
What is this Stuff? I Dunno, But Let’s Take it Anyway!
Recent research shows many people are exceeding recommended daily dosage of NSAIDs, and are unwittingly ingesting products they do not realize are NSAIDs, sometimes while consuming other NSAIDs. A survey of 1,326 US ibuprofen users found 37% also took non-ibuprofen NSAIDs and that most did not recognize all relevant products they’d taken as NSAIDs. Exceeding the recommended daily limit occurred among 11% of ibuprofen users, 4% of users for other NSAIDs, and on 9.1% of NSAID usage days. The results suggested that 15% of adult ibuprofen users in the US exceed the maximum recommended daily dose of 1 or more over‐ the‐counter or prescription NSAIDs during any week in which they use ibuprofen (Kaufman et al, 2018).
In a finding that would be comical if it were not in fact a depressing reminder of just how dumb so many Homo sapiens are, many respondents believed they could choose their own NSAID dose regardless of label directions. This was especially so for those who exceeded the daily limit.
A survey of over 800 Parkrun UK participants found a high use of NSAIDs during the past 12 months (88%). Ibuprofen was the most commonly used NSAID (81%). A third of runners had experienced an adverse drug reaction associated with NSAIDs, usually gastrointestinal. Half of runners used NSAIDs with no advice, with patient information leaflets the most common source for those that had advice. Eleven percent of respondents said they were not aware of any adverse drug reactions that could emanate from NSAID use (Rosenbloom et al, 2020).
Can Someone Round Up Influencers and Stick Them in a Remote Outback Enclosure, Please?
Influencers, aka marketing-savvy grifters, have been pushing NSAIDs through their social media platforms. As part of a social media campaign using the hashtag #advilpartner, a number of shameless characters accepted money to pimp Haleon’s Advil, a US brand of pain-relieving medication that features ibuprofen as its primary ingredient.
“Pain is inevitable… nothing works faster … Advil keeps me going,” reads the text on a February 2025 Instagram reel by hybrid athlete MaKayla Bassi (100,000 followers) while she was running on a track.
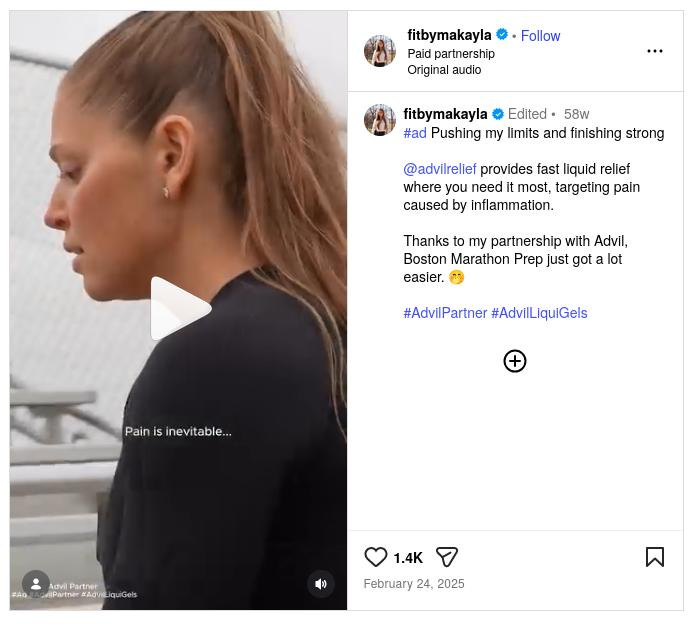
Someone’s talking scheissen, because pain is not inevitable when you train. Sure, some degree of discomfort is par for the course when you’re training hard. The solution is to grit your teeth, harden the f**k up, and realize that achieving worthwhile goals – like not being a mushy lard-ass – often requires effort, resilience and fortitude. Not goddamn drugs.
However, if Bassi is truly experiencing unavoidable pain while she’s running, she needs to stop masquerading as an expert and seek the help of a real professional immediately. Experiencing pain so debilitating while running that it requires drug use to ‘keep you going’ is not normal. Either Bassi is 1) ignoring a serious injury or medical condition, 2) hopelessly soft, and/or 3) bullshitting for monetary gain.
“[Advil] works in under 30 minutes, helping me power through and have a good workout,” running influencer Mica Wood (107,000 followers) shared in a similar video, who used the “menstrual pain” angle to justify her paid promotion of Advil.
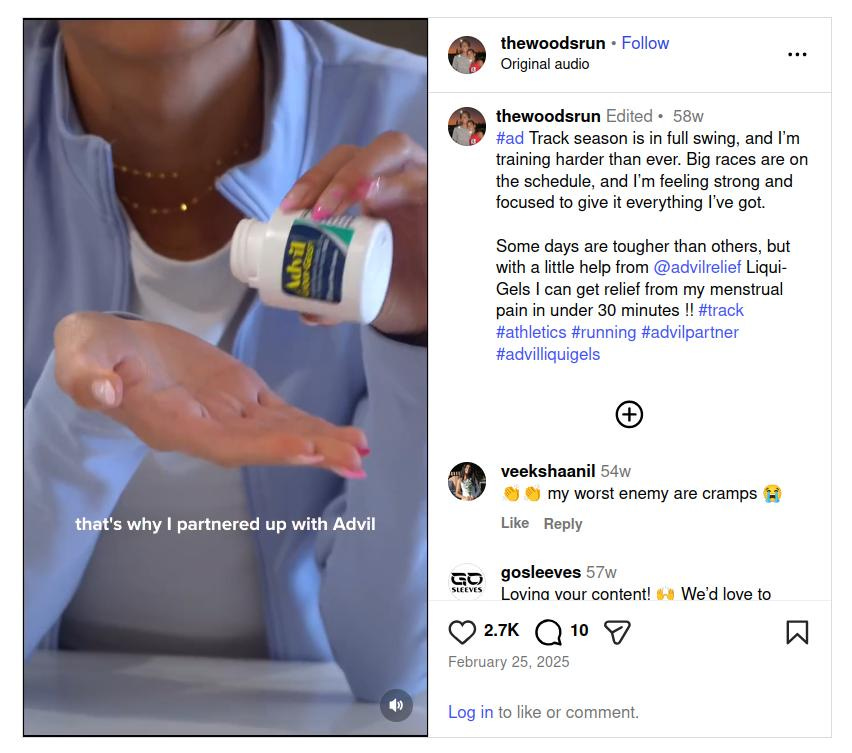
Ginger has also shown efficacy for period pain (Daily et al, 2015; Chen et al, 2016), and numerous small studies have found it to be just as effective as ibuprofen (and mefenamic acid) for treating menstrual pain (Ozgoli et al, 2009; Halder, 2012; Jenabi, 2013; Shirvani et al, 2015; Adib Rad et al, 2018). It’s a finding calling out for larger trials, but don’t expect Big Pharma or its stooges in Big Gov to fund research that will likely show a simple herb more efficacious in treating a common malady than one of the world’s most popular drug classes.
And, of course, don’t expect Wood to start pimping the zingy herb unless ginger farmers start fronting up with some cash.
“Wanna know my secret weapon as I train for my next race?” running influencer Amy Haas (138,000 followers) said in a now-deleted video reel.
Not really Amy, but go on.
“Running comes with lots of aches and pains...which is why I’m working with Advil.”
Sure. It wouldn’t be because they’re sending you money to pimp this porqueria, would it now?

After receiving thoroughly-deserved backlash, Haas edited another post to include a disclaimer that combining Advil and running can result in further damage to both kidneys and muscles.
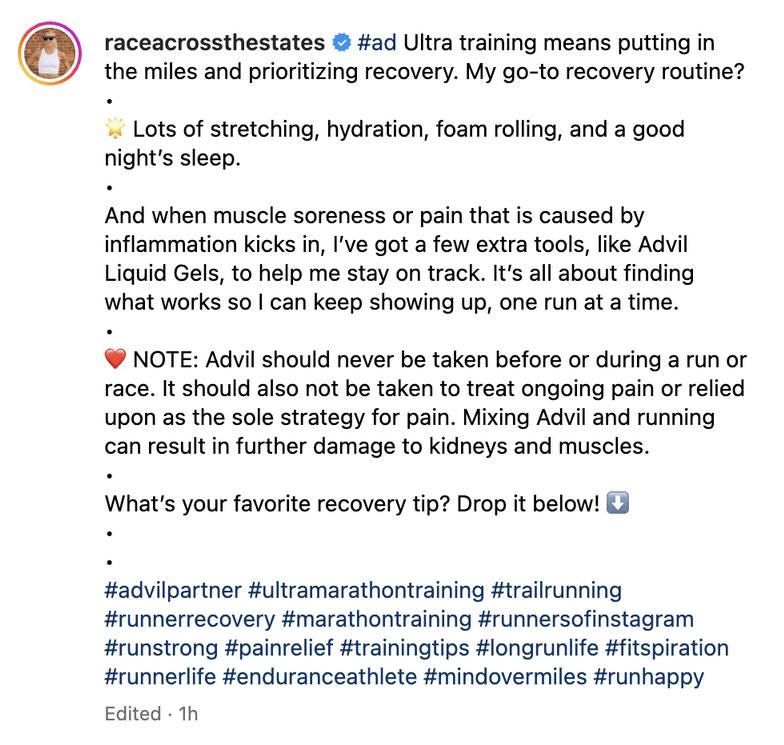
Some joker who calls herself “Dr Lia” (real name Amelia Cruz, 152K followers) wrote on Instagram:
“🔐 Real strength doesn’t come from pushing through pain but recognizing and treating it to find relief. Advil is launching ✨The Strength Shift✨, a bold movement that’s redefining what real strength looks like: confronting and treating pain, not pushing through it🚫”
If that sounds like a load of convoluted horseshit, allow me to decipher what she’s really trying to say:
“I’m pimping this crap because Haleon are paying me!”
You’ll note that all the aforementioned Advil spruikers are female. This is not by coincidence; females are more likely to report pain and use NSAIDs than males, which makes them a lucrative target group.
Oh, and females are also more likely to experience adverse drug reactions from NSAIDs, something the Advil-pimping sistahood kinda sorta forgets to mention in its paid social media promotions (Rogers et al, 2024).
You go girls!
Straight to ethics classes.
Conclusion
From the research I’ve discussed above, we can deduce several key points about NSAIDs.
The first is that many grown adults are, quite frankly, too dumb to be taking these drugs. If you can’t identify a class of drugs and don’t fully comprehend what they do, you really shouldn’t be self-medicating with them. Taking potentially toxic analgesics when you are not experiencing any pain, or “very mild/mild” pain, really is quite stupid. Furthermore, if the labeling for a drug stipulates not to exceed a specific dose, but you do so anyway because, hey, you’re special and think you can do what you like without consequence, then you need to go back to playing with building blocks and coloring books. Use that as a starting point to build up your IQ. Until you evolve into something resembling an intelligent and mature adult, forget about drugs, which are a risky proposition at the best of times. Adopting la-la land attitudes towards drug consumption is just asking for trouble.
As for taking drug advice from social media influencers, a group infamous for their lack of ethics, morals, and mental stability … do I really need to explain why this is a bad idea?
The second observation is that, for many if not most people who use them, NSAIDs are useless and likely counterproductive. Forking over money for something that not only has no positive effect on healing but may in fact hinder it is a lose-lose proposition. Prostaglandin release is important for optimal muscle healing, and NSAIDs inhibit prostaglandins via their ‘anti-inflammatory’ effects.
The third clear take-home message is that these drugs are more dangerous than what people realize. You can sit there and object that, hey, given the squillions of people that take these drugs, the risk isn’t that great.
I counter that when these drugs offer very little benefit for most people but the very real possibility of serious bleeding, renal and cardiovascular events, they possess an unacceptable risk:reward profile.
For these reasons, I rate NSAIDs 5 steaming piles of horseshit. When it comes to recovering from training injuries, I would (and do) opt for natural, non-NSAID (and non-acetaminophen) modalities.
As for those experiencing mild-to-zero pain, if I were a doctor my go-to recommendation would be a daily dose of Harden The F**k Up™. HTFU™ is an amazing therapy and readily available without prescription; side effects include increased accountability, reduced self-pity and realizing how pathetically soft you’ve been. I mean, kids are starving in Yemen and getting their limbs blown off in Gaza, and you’re taking drugs for no or “very mild” pain?!?
Ugh. First world problems.
Anyhow, have some ginger biscotti.
Ciao,
Anthony.
Hi Joseph,
did you any actual research into this issue yourself, or is your contention that the bleeding risk from aspirin is a "myth" derived solely from articles like the Robert Yoho one you linked to?
Unlike most other NSAIDs, aspirin does indeed have definite beneficial use cases - for example, in people who have just had an ischemic heart attack or stroke, it can literally be a lifesaver and is routinely administered in hospitals for that purpose.
The claim that there is a surreptitious campaign to suppress aspirin and crush Bayer - whose major shareholders include BlackRock and Vanguard - is fanciful given that aspirin has long been openly and routinely pimped as a "one a day" preventive against CVD.
As noted, if you end up in the emergency unit because of an ischemic event, the doctors are very likely to administer aspirin - not ibuprofen or acetaminophen.
The Yoho article is full of fantasmagorical claims, and when I click on the links to verify, those links are usually broken.
For example, his assertion that "The Harvard Nurses and the FULL studies showed that taking a single uncoated 325 mg aspirin daily for several years reduces the risk of cancer by over 50%."
He later claims, without any reference, that Canadian research showed a 60% reduction in cancer risk.
This figure further rises as high as 73% (for esophageal cancer), again using no or broken links as references.
The truth is that numerous studies have indeed found longer-term (10+ years) use of aspirin is associated with reduced cancer risk, but not of the magnitude Yoho is claiming.
And the bleeding risk is not a myth, it is very real.
Zheng et al analysed 13 trials, incorporating over 164,000 participants with no known CVD risk, and found the absolute risk of bleeding was greater than the absolute risk reduction in composite cardiovascular outcome.
https://jamanetwork.com/journals/jama/fullarticle/2721178
Meaning, aspirin is not a 'wonder drug' (I'm immediately wary of anyone who uses that term) but a pharmaceutical whose benefits have to be weighed against the risks. This is what intelligent doctors are supposed to do - review individual patient data, and decide if the drug is a good risk:reward proposition for that patient. Not write internet articles declaring a sinister suppression and encouraging all and sundry to start taking it pronto, using fantastic RR figures that cannot be verified because the links are broken.
Hastie et al also studied pregnant women and found aspirin users had a higher incidence of intrapartum bleeding, postpartum hemorrhage, postpartum hematoma and neonatal intracranial hemorrhage.
https://www.ajog.org/article/S0002-9378(20)30737-7/abstract
I don't know about you, but I wouldn't want to jeopardize the life of my significant other nor Junior A-Train because of a bunch of 'wonder drug' dogma.
Cheers,
A.
Bjorkman D. J. (1998). The effect of aspirin and nonsteroidal anti-inflammatory drugs on prostaglandins. American Journal of Medicine, 105(1B), 8S–12S. https://doi.org/10.1016/s0002-9343(98)00069-2
Bondesen, B. A., Mills, S. T., & Pavlath, G. K. (2006). The COX-2 pathway regulates growth of atrophied muscle via multiple mechanisms. American Journal of Physiology. Cell physiology, 290(6), C1651–C1659. https://doi.org/10.1152/ajpcell.00518.2005
Brewer, C.B., & Waddell, D. (2012). The Role of Prostaglandin F 2α in Skeletal Muscle Regeneration. Journal of Trainology, 1, 45-52. https://doi.org/10.17338/trainology.1.2_45
Burd, N. A., Dickinson, J. M., Lemoine, J. K., Carroll, C. C., Sullivan, B. E., Haus, J. M., Jemiolo, B., Trappe, S. W., Hughes, G. M., Sanders, C. E., Jr, & Trappe, T. A. (2010). Effect of a cyclooxygenase-2 inhibitor on postexercise muscle protein synthesis in humans. American journal of physiology. Endocrinology and metabolism, 298(2), E354–E361. https://doi.org/10.1152/ajpendo.00423.2009
Chen, C. X., Barrett, B., & Kwekkeboom, K. L. (2016). Efficacy of Oral Ginger (Zingiber officinale) for Dysmenorrhea: A Systematic Review and Meta-Analysis. Evidence-based complementary and alternative medicine : eCAM, 2016, 6295737. https://doi.org/10.1155/2016/6295737
Chesley, A., MacDougall, J. D., Tarnopolsky, M. A., Atkinson, S. A., & Smith, K. (1992). Changes in human muscle protein synthesis after resistance exercise. Journal of applied physiology, 73(4), 1383–1388. https://doi.org/10.1152/jappl.1992.73.4.1383
Ciuciu, A., Mulholland, C., Bozzi, M. A., Frymoyer, C. C., Cavinatto, L., Yaron, D., Harwood, M. I., Close, J. D., Mehallo, C. J., & Tomlinson, R. E. (2024). Regular Nonsteroidal Anti-Inflammatory Drug Use Increases Stress Fracture Risk in the General Population: A Retrospective Case-Control Study. Advances in orthopedics, 2024, 7933520. https://doi.org/10.1155/2024/7933520
Daily, J. W., Zhang, X., Kim, D. S., & Park, S. (2015). Efficacy of Ginger for Alleviating the Symptoms of Primary Dysmenorrhea: A Systematic Review and Meta-analysis of Randomized Clinical Trials. Pain medicine, 16(12), 2243–2255. https://doi.org/10.1111/pme.12853
Davis, A., & Robson, J. (2016). The dangers of NSAIDs: look both ways. British journal of general practice : the journal of the Royal College of General Practitioners, 66(645), 172–173. https://doi.org/10.3399/bjgp16X684433
Dhikav, V., Gupta, S., & Anand, K. S. (2008). Erectile dysfunction induced probably by celecoxib. Pharmacoepidemiology and drug safety, 17(2), 211–212. https://doi.org/10.1002/pds.1539
Funk CD (2001) Prostaglandins and leukotrienes: advances in eicosanoid biology. Science, 294, 1871–1875. https://doi.org/10.1126/science.294.5548.1871
García-Martínez, O., De Luna-Bertos, E., Ramos-Torrecillas, J., Manzano-Moreno, F. J., & Ruiz, C. (2015). Repercussions of NSAIDS drugs on bone tissue: the osteoblast. Life sciences, 123, 72–77. https://doi.org/10.1016/j.lfs.2015.01.009
Gorfinkel, L. R., Hasin, D., Saxon, A. J., Wall, M., Martins, S. S., Cerdá, M., Keyes, K., Fink, D. S., Keyhani, S., Maynard, C. C., & Olfson, M. (2022). Trends in Prescriptions for Non-opioid Pain Medications Among U.S. Adults With Moderate or Severe Pain, 2014-2018. Journal of pain, 23(7), 1187–1195. https://doi.org/10.1016/j.jpain.2022.01.006
Haerdtlein, A., Debold, E., Rottenkolber, M., Boehmer, A. M., Pudritz, Y. M., Shahid, F., Gensichen, J., & Dreischulte, T. (2023). Which Adverse Events and Which Drugs Are Implicated in Drug-Related Hospital Admissions? A Systematic Review and Meta-Analysis. Journal of clinical medicine, 12(4), 1320. https://doi.org/10.3390/jcm12041320
Halder A. (2012). Effect of progressive muscle relaxation versus intake of ginger powder on dysmenorrhoea amongst the nursing students in Pune. Nursing journal of India, 103(4), 152–156.
Jenabi E. (2013). The effect of ginger for relieving of primary dysmenorrhoea. JPMA: Journal of the Pakistan Medical Association, 63(1), 8–10.
Karha, J., & Topol, E. J. (2004). The sad story of Vioxx, and what we should learn from it. Cleveland Clinic journal of medicine, 71(12), 933–939. https://doi.org/10.3949/ccjm.71.12.933
Kaufman, D. W., Kelly, J. P., Battista, D. R., Malone, M. K., Weinstein, R. B., & Shiffman, S. (2018). Exceeding the daily dosing limit of nonsteroidal anti-inflammatory drugs among ibuprofen users. Pharmacoepidemiology and drug safety, 27(3), 322–331. https://doi.org/10.1002/pds.4391
Kongkaew, C., Noyce, P. R., & Ashcroft, D. M. (2008). Hospital admissions associated with adverse drug reactions: a systematic review of prospective observational studies. The Annals of pharmacotherapy, 42(7), 1017–1025. https://doi.org/10.1345/aph.1L037
Krentz, J. R., Quest, B., Farthing, J. P., Quest, D. W., & Chilibeck, P. D. (2008). The effects of ibuprofen on muscle hypertrophy, strength, and soreness during resistance training. Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme, 33(3), 470–475. https://doi.org/10.1139/H08-019
Küster, M., Renner, B., Oppel, P., Niederweis, U., & Brune, K. (2013). Consumption of analgesics before a marathon and the incidence of cardiovascular, gastrointestinal and renal problems: a cohort study. BMJ open, 3(4), e002090. https://doi.org/10.1136/bmjopen-2012-002090
Li, T., Wu, C., Fu, F., Qin, F., Wei, Q., & Yuan, J. (2018). Association between use of aspirin or non-aspirin non-steroidal anti-inflammatory drugs and erectile dysfunction: A systematic review. Medicine, 97(28), e11367. https://doi.org/10.1097/MD.0000000000011367
Lilja, M., Mandić, M., Apró, W., Melin, M., Olsson, K., Rosenborg, S., Gustafsson, T., & Lundberg, T. R. (2018). High doses of anti-inflammatory drugs compromise muscle strength and hypertrophic adaptations to resistance training in young adults. Acta physiologica, 222(2), 10.1111/apha.12948. https://doi.org/10.1111/apha.12948
Lim, J. S., Han, S., Kim, J. S., Kim, S., & Oh, B. (2025). Effects of NSAIDs on Early CKD Development: A 10-Year Population-Based Study Using the Korean Senior Cohort. Drugs & aging, 42(10), 953–961. https://doi.org/10.1007/s40266-025-01239-9
Lipman, G. S., Shea, K., Christensen, M., Phillips, C., Burns, P., Higbee, R., Koskenoja, V., Eifling, K., & Krabak, B. J. (2017). Ibuprofen versus placebo effect on acute kidney injury in ultramarathons: a randomised controlled trial. Emergency medicine journal : EMJ, 34(10), 637–642. https://doi.org/10.1136/emermed-2016-206353
Mackey, A. L., Kjaer, M., Dandanell, S., Mikkelsen, K. H., Holm, L., Døssing, S., Kadi, F., Koskinen, S. O., Jensen, C. H., Schrøder, H. D., & Langberg, H. (2007). The influence of anti-inflammatory medication on exercise-induced myogenic precursor cell responses in humans. Journal of applied physiology, 103(2), 425–431. https://doi.org/10.1152/japplphysiol.00157.2007
Mallinson, J. E., Taylor, T., Constantin-Teodosiu, D., Constantin, D., Franchi, M. V., Auer, D., & Greenhaff, P. L. (2025). NSAID ingestion augments training-induced muscle hypertrophy and differentially affects muscle mRNA expression, but not strength gains, in trained men. The Journal of physiology, 10.1113/JP289542. Advance online publication. https://doi.org/10.1113/JP289542
Mikkelsen, U. R., Langberg, H., Helmark, I. C., Skovgaard, D., Andersen, L. L., Kjaer, M., & Mackey, A. L. (2009). Local NSAID infusion inhibits satellite cell proliferation in human skeletal muscle after eccentric exercise. Journal of Applied Physiology, 107(5), 1600–1611. https://doi.org/10.1152/japplphysiol.00707.2009
Ozgoli, G., Goli, M., & Moattar, F. (2009). Comparison of effects of ginger, mefenamic acid, and ibuprofen on pain in women with primary dysmenorrhea. Journal of alternative and complementary medicine, 15(2), 129–132. https://doi.org/10.1089/acm.2008.0311
Paulsen, G., Egner, I. M., Drange, M., Langberg, H., Benestad, H. B., Fjeld, J. G., Hallén, J., & Raastad, T. (2010). A COX-2 inhibitor reduces muscle soreness, but does not influence recovery and adaptation after eccentric exercise. Scandinavian journal of medicine & science in sports, 20(1), e195–e207. https://doi.org/10.1111/j.1600-0838.2009.00947.x
Petersen, S. G., Miller, B. F., Hansen, M., Kjaer, M., & Holm, L. (2011a). Exercise and NSAIDs: effect on muscle protein synthesis in patients with knee osteoarthritis. Medicine and science in sports and exercise, 43(3), 425–431. https://doi.org/10.1249/MSS.0b013e3181f27375
Petersen, S. G., Beyer, N., Hansen, M., Holm, L., Aagaard, P., Mackey, A. L., & Kjaer, M. (2011b). Nonsteroidal anti-inflammatory drug or glucosamine reduced pain and improved muscle strength with resistance training in a randomized controlled trial of knee osteoarthritis patients. Archives of physical medicine and rehabilitation, 92(8), 1185–1193. https://doi.org/10.1016/j.apmr.2011.03.009
Phillips, S. M., Tipton, K. D., Aarsland, A., Wolf, S. E., & Wolfe, R. R. (1997). Mixed muscle protein synthesis and breakdown after resistance exercise in humans. American journal of physiology, 273(1 Pt 1), E99–E107. https://doi.org/10.1152/ajpendo.1997.273.1.E99
Reynolds, J. F., Noakes, T. D., Schwellnus, M. P., Windt, A., & Bowerbank, P. (1995). Non-steroidal anti-inflammatory drugs fail to enhance healing of acute hamstring injuries treated with physiotherapy. South African medical journal = Suid-Afrikaanse tydskrif vir geneeskunde, 85(6), 517–522.
https://www.researchgate.net/publication/15545790_Non-steroidal_anti-inflammatory_drugs_fail_to_enhance_healing_of_acute_hamstring_injuries_treated_with_physiotherapy
Roberts, B. M., Sczuroski, C. E., Caldwell, A. R., Zeppetelli, D. J., Smith, N. I., Pecorelli, V. P., Gwin, J. A., Hughes, J. M., & Staab, J. S. (2024). NSAIDs do not prevent exercise-induced performance deficits or alleviate muscle soreness: A placebo-controlled randomized, double-blinded, cross-over study. Journal of science and medicine in sport, 27(5), 287–292. https://doi.org/10.1016/j.jsams.2024.02.002
Rogers, P., Wang, D., Lu, Z., & Lyn-Cook, B. (2024). Identifying Vulnerabilities to NSAID Adverse Events in the U.S. Population: An Analysis of Preexisting Conditions and Sex. Journal of women’s health, 33(7), 901–907. https://doi.org/10.1089/jwh.2023.1039
Rosenbloom, C. J., Morley, F. L., Ahmed, I., & Cox, A. R. (2020). Oral non-steroidal anti-inflammatory drug use in recreational runners participating in Parkrun UK: Prevalence of use and awareness of risk. The International journal of pharmacy practice, 28(6), 561–568. https://doi.org/10.1111/ijpp.12646
Shirvani, M. A., Motahari-Tabari, N., & Alipour, A. (2015). The effect of mefenamic acid and ginger on pain relief in primary dysmenorrhea: a randomized clinical trial. Archives of gynecology and obstetrics, 291(6), 1277–1281. https://doi.org/10.1007/s00404-014-3548-2
Trappe, T. A., Fluckey, J. D., White, F., Lambert, C. P., & Evans, W. J. (2001). Skeletal muscle PGF(2)(alpha) and PGE(2) in response to eccentric resistance exercise: influence of ibuprofen acetaminophen. Journal of clinical endocrinology and metabolism, 86(10), 5067–5070. https://doi.org/10.1210/jcem.86.10.7928
Trappe, T. A., White, F., Lambert, C. P., Cesar, D., Hellerstein, M., & Evans, W. J. (2002). Effect of ibuprofen and acetaminophen on postexercise muscle protein synthesis. American journal of physiology. Endocrinology and metabolism, 282(3), E551–E556. https://doi.org/10.1152/ajpendo.00352.2001
Trappe, T. A., Carroll, C. C., Dickinson, J. M., LeMoine, J. K., Haus, J. M., Sullivan, B. E., Lee, J. D., Jemiolo, B., Weinheimer, E. M., & Hollon, C. J. (2011). Influence of acetaminophen and ibuprofen on skeletal muscle adaptations to resistance exercise in older adults. American journal of physiology. Regulatory, integrative and comparative physiology, 300(3), R655–R662. https://doi.org/10.1152/ajpregu.00611.2010
Van Wijck, K., Lenaerts, K., Van Bijnen, A. A., Boonen, B., Van Loon, L. J., Dejong, C. H., & Buurman, W. A. (2012). Aggravation of exercise-induced intestinal injury by Ibuprofen in athletes. Medicine and science in sports and exercise, 44(12), 2257–2262. https://doi.org/10.1249/MSS.0b013e318265dd3d
Vassallo, F., Martinelli, M., Varcamonti, L., & Buono, P. (2025). Adverse reactions to acetaminophen and ibuprofen in pediatric patients: a narrative review. Italian journal of pediatrics, 51(1), 295. https://doi.org/10.1186/s13052-025-02135-z
Wheatley, B. M., Nappo, K. E., Christensen, D. L., Holman, A. M., Brooks, D. I., & Potter, B. K. (2019). Effect of NSAIDs on Bone Healing Rates: A Meta-analysis. Journal of the American Academy of Orthopaedic Surgeons, 27(7), e330–e336. https://doi.org/10.5435/JAAOS-D-17-00727