Before continuing on with this exposé of anti-depressants, I want to reiterate a plea I made in Part 1:
Namely, if you are feeling suicidal, please confide in someone.
I reiterate this plea because, since the last instalment in this series, there has been a tragic development. Two months ago, one of my younger cousins committed suicide. He was only 32. He’d had some major challenges in life, but no-one in the family had any inkling of what he was contemplating. He was a great kid, and now he’s gone, leaving behind a heartbroken family and shocked, saddened relatives. I know I speak for the rest of my relatives when I say that if any of us had known what he was contemplating, we would have done whatever we possibly could have to help him.
So please – if you are severely depressed and/or feeling suicidal, don’t suffer your mental torment in silence.
Call a friend, call a family member, call a help line, call a counsellor, teacher, minister … someone.
Keep trying until you find a sympathetic, caring ear. As Bev Aisbett noted in her book Taming the Black Dog: A Guide to Overcoming Depression:
“When choosing your confidante, keep in mind that not everyone’s good at it! Don’t be disheartened if someone can’t handle it; the important thing is that you have reached out! Keep reaching out until you get the help you need.”
And if you are currently taking antidepressants, remember that they are well-documented to increase the risk of suicidal ideation and suicide attempts. It might not really be you that wants to end it all - it could be the drugs. Don’t let that toxic junk do your talking for you.
Life is already way too short.
So with that, let’s return to the topic at hand: SSRI antidepressants and the increased suicide risk they bring.
Getting Real About the Drug Companies
The current official consensus on SSRI anti-depressants and suicide risk is that they only pose an increased risk in adolescents (which has done little to stop doctors routinely prescribing these drugs to adolescents).
In adults, especially those over 65, the ‘experts’ tell us these drugs actually reduce risk. If you jump onto PubMed or your favourite search engine and run a search for “SSRI suicide risk”, you’ll find no shortage of articles, reviews and meta-analyses peddling this official line.
It is important to note these so-called experts are invariably either drug company-sponsored researchers, or investigators from outfits like the US Food and Drug Administration (FDA), whose overly cosy relationship with Big Pharma has been well documented.
And so it’s time to address the elephant in the room that all these 'experts' continue to conveniently ignore:
Namely, drug companies are often criminal enterprises that place profits far above human health. As such, they have no qualms about manipulating and falsifying research data in order to make their drugs appear safer and more effective than what they really are.
Their shady tactics go far beyond just data manipulation, however. In their relentless mission to boost profits, drug companies have engaged in everything from using skanky strippers to corruptly fixing court cases.
Drug Company Sleaze
Already drug-addled countries like the United States and Australia are now currently in the midst of a prescription opioid drug abuse epidemic. Opioid drugs have long since established themselves as one very nasty class of drugs: Highly dangerous and highly addictive.
Among the best known of these highly addictive drugs is OxyContin, made by Purdue Pharma. Purdue’s owners, the wealthy Sackler family, now stand accused of conducting a concerted campaign to mislead doctors and patients about the dangers of the powerful painkiller.
Purdue sales representatives focused on doctors who were high-volume opioid prescribers, as well as inexperienced providers and physicians who knew little about pain management. They were encouraged to prescribe increasingly higher doses for longer stretches of time, according to court filings. Sales representatives could earn tens of thousands of dollars in bonuses and were rewarded with trips to tropical islands.
Since OxyContin came on the market, more than 200,000 Americans have died of overdoses related to prescription opioids. As reports of overdoses grew the then-president of Purdue, Richard Sackler, urged the company to blame the patients. “We have to hammer on abusers in every way possible,” he wrote in a 2001 email disclosed in documents filed in the Massachusetts case. “They are the culprits and the problem. They are reckless criminals.”
If the courts decide Purdue was in fact the recklessly criminal entity, it could cost it dearly: The company is now being pounded with lawsuits from all angles.
 Richard Sackler. Promoted excessive prescribing of OxyContin, then labelled addicts the “culprits” and “reckless criminals.”
Richard Sackler. Promoted excessive prescribing of OxyContin, then labelled addicts the “culprits” and “reckless criminals.”
Another recent US case highlighting just how low drug companies will stoop is the federal racketeering trial against executives from Insys. Prosecutors allege that the executives used bribes and kickbacks to get doctors to prescribe the company’s powerful and addictive fentanyl spray, called Subsys. Fentanyl is an especially problematic opioid drug that was intended only for cancer patients experiencing pain not alleviated by other medications. In 2016 alone, fentanyl and fentanyl analogues accounted for 20,100 of the 64,000 drug overdoses that killed people in the United States.
The former Insys executives are also accused of misleading and defrauding health insurance companies who covered the drug for patients who did not need it. A congressional investigation in 2017 concluded Insys sales representatives bluntly lied and tricked insurers to do just that — and investigators released the tapes to prove it.
This kind of corruption is par for the course for drug companies. But in its insatiable quest for profits, Insys decided to get extra creative.
One of the executives on trial is Insys sales director, Sunrise Lee. You might think that to become a sales director at a pharmaceutical company, one would need a proven sales track record in the pharmaceutical industry. Lee's previous work history, however, did not involve pharmaceutical sales; rather, it entailed rubbing her private bits against a pole. That's right: Lee is a former stripper, and had no experience in the pharmaceutical industry before working at Insys. According to Bloomberg, Insys whistle-blowers quoted former Vice President of Sales Alec Burlakoff as saying that “doctors really enjoyed spending time with [Lee]” and that she was “more of a closer.”
Right.
Former Insys sales representative Holly Brown testified that Lee played up her sexuality during sales pitches. She dressed in a “sexually suggestive manner’’ and “showed more cleavage than the average sales rep,’’ Brown said.
Brown further testified that in mid-2012 she, Lee, and other sale representatives took one Dr. Paul Madison to a popular Chicago nightclub called the Underground after a dinner event sponsored by Insys. Brown said her superiors had told her to focus on Dr. Madison, who was known for prescribing a lot of opioids. She described his office as a “shady pill mill” in a “dingy strip mall in a not-so-nice area of town.”
Brown recalled an encounter she witnessed that night between Lee and Madison, saying, “She was sitting on his lap, kind of bouncing around, and he had his hands all over her chest.’’
As well as allowing him to feel up the breasts of its sales director, prosecutors reported that Insys paid Madison at least $70,800 in “speaker fees.”
 Stripper-turned-pharmaceutical sales director, Sunrise Lee.
Stripper-turned-pharmaceutical sales director, Sunrise Lee.
When a doctor prescribes medication, patients assume he is doing so because that medication has been carefully tested and shown to be effective for their condition. If they knew that dubious sales techniques, fraud, bribes and even sleazy lap dances had played a key role in the prescribing decision, they might be a little more questioning.
Big Corruption
Companies like Insys and Purdue are more Mid Pharma than Big Pharma. Their shady antics begin to look like amateur hour stuff when compared to that of the true behemoths of the pharmaceutical industry.
Without question, the best-known anti-depressant is the infamous SSRI Prozac (fluoxetine), made by Eli Lilly and Co., the global pharmaceutical company headquartered in Indianapolis, Indiana. With some 40,000 employees and over $24 billion annually in sales, Eli Lilly ranks among the largest pharmaceutical companies in the world.
According to its website, "Lilly unites caring with discovery to create medicines that make life better for people around the world."
Eli Lilly has a rather unique approach to “caring” and making “life better for people”. Thanks to the legal participation of folks like Dr Peter Breggin, we now know its strategies for attaining this lofty goal included fixing a jury trial and misrepresenting research data to dramatically downplay the suicide risk of Prozac.
Dr Breggin Uncovers the Lies Behind SSRIs
Peter R. Breggin MD is a Harvard-trained psychiatrist and former consultant at the National Institute of Mental Health (NIMH) and for the Federal Aviation Administration (FAA). He has tirelessly worked for decades to reform the mental health field. He first became significantly involved in the medical-legal arena in the early 1970s as a medical expert in a trial that helped stop the resurgence of lobotomy and other barbaric forms of psychosurgery. In the early 1990s, he was asked by a consortium of attorneys to become the medical expert in a large series of combined lawsuits against Eli Lilly and Co. concerning Prozac. He continues to be consulted as an expert in various legal actions involving SSRIs, including Prozac, Zoloft, Paxil, Celexa, and Luvox.
When a plaintiff sets out to sue a corporation (or a hopelessly corrupt government entity), one of the first steps is to file a motion for discovery. If approved by the judge, the company or corrupt government entity is then obliged to release all of its relevant records to the plaintiff.
As Breggin recounts in his must-read The Antidepressant Fact Book: What Your Doctor Won't Tell You About Prozac, Zoloft, Paxil, Celexa, And Luvox, after the plaintiff lawyers in the early Prozac suits filed their motions for discovery, he ended up with a mini-mountain of internal documents. Over the next year, as he pored through these documents, he began developing the scientific basis for hundreds of suits against the company revolving around charges Prozac caused violence, suicide, and psychosis.
While scrutinizing these internal documents, Dr Breggin found enormously important information Eli Lilly purposely withheld from the FDA, the medical profession and the public. He also found innumerable instances of unsound and unethical scientific practices. He discovered, for example, that Eli Lilly systematically mislabelled many adverse reactions to Prozac in clinical trials so they would be hidden from view. A suicide by a clinical trial participant taking Prozac would often be filed away under the heading “no drug effect” or “depression.” When the FDA or anyone else then examined the rates of suicide on patients taking Prozac, those patient suicides would never be counted, because a computer search would identify them as “no drug effect” or “depression” rather than “suicide.”
Which brings us to a key flaw of all these reviews and meta-analyses purporting to have found no increased suicide risk among adult clinical trial participants taking SSRIs:
They are based on incomplete and corrupted data.
Dr Breggin documented how the company manipulated its own clinical trials so that Prozac would come out looking better and less harmful than it really was. He found secret studies by the company showing increased rates of overstimulation and suicide attempts on Prozac during clinical trials. He also examined FDA reports and correspondence about Prozac that the government agency refused to disclose in response to freedom of information requests.
Due in no small part to Dr Breggin’s findings, Eli Lilly has since been sued hundreds of times for negligence and fraudulence in the testing, development, and marketing of Prozac. Although Dr Breggin was directly involved in only a fraction of those cases, his research probably provided the basis for most of them. Eli Lilly has secretly and quietly settled many, if not most, of these suits. And you can be sure as heck that Eli Illy doesn’t include the results of these cases in its promotional efforts to doctors and the public.
The (Jury Trial) Fix Is In
When he began working on the combined Prozac suits in the early 1990s, Breggin collaborated primarily with two lawyers, the second of which was Leonard Ring, a highly regarded Chicago attorney.
Unfortunately, Ring died in 1994 and was replaced by Paul Smith, a Dallas attorney. “Smith,” recounts Breggin, “became the leader of the entire legal assault on Eli Lilly and Co. and prepared to bring the first case to court.”
Smith’s behaviour was puzzling from the outset. Recalls Breggin: “Unlike every other attorney who has ever hired me, Smith spent almost no time going over the case with me. I was left on my own to evaluate enormous amounts of material and to develop the substance of my future testimony.”
Smith's bizarre antics continued after Breggin arrived in Louisville, Kentucky, for the first Prozac case to go to court. The case revolved around Joseph Wesbecker, a Kentucky man who, after being prescribed Prozac, wounded twenty and killed 8 of his former workmates before killing himself.
Upon arriving in Louisville, Breggin was shocked to find out that Smith had sequestered him in a hotel miles away from where Smith and the other attorneys were staying. In other cases, attorneys put him in the same hotel with them to make it easier to review facts and issues together.
At this point, Breggin and his wife became suspicious. "My suspicions turned into grave concerns when Smith refused to discuss my upcoming testimony with me," says Breggin. "He wanted me to get on the stand and respond to whatever he happened to ask me. Attorneys in huge suits like this usually spend many hours and even days going over the testimony in advance with the expert."
Breggin then found out Smith had kept many relevant documents from him, including a detailed, color-coded chart about Joseph Wesbecker's childhood that had been created by the drug company from its investigations. When Breggin asked for a copy of the biographical chart, Smith refused, and only gave in after Breggin vigorously demanded it. Smith also continued to refuse to discuss Breggin's testimony with him.
When Breggin took the stand, much of Eli Lilly's cross-examination of him was based on the childhood chart Smith had unsuccessfully tried to keep from him. Breggin had obviously been set up to look foolish and make Eli Lilly look good, but this diabolical plan went awry because Breggin found the sequestered chart and was able to answer the questions in extensive detail.
As Breggin sat in the stand, Smith refused to ask him about some of the most important discoveries he’d made when scouring through the Eli Lilly documents. For example, he discovered the Prozac label originally listed “depression” as a commonly reported adverse reaction to Prozac. Breggin had showed Smith a copy of the label changes showing how it had been scratched out at the last minute. Eli Lilly knew its so-called 'anti-depressant' drug could cause depression and yet allowed the fact to be removed from the label, but Smith refused to let Breggin discuss this striking new evidence.
Smith even allowed Eli Lilly to attack Breggin during their summation to the jury without making any attempt to rebut their criticism in his own summation.
Thanks to Smith’s obstruction, Breggin was unable to relay the full extent of Eli Lilly’s malfeasance to the jury. As a result, the jury came back in favor of the drug company by a divided 9–3 vote. One more vote against Eli Lilly would have hung the jury and created a PR nightmare for Prozac.
The presiding judge John W. Potter, however, was no dummy. Soon after the trial, he concluded Eli Lilly had rigged it. The company had secretly given a huge sum of money to Smith and his clients in return for Smith presenting a watered-down case to the jury. "No wonder Smith tried to stifle my testimony," says Breggin. "In return for a mammoth advance settlement, he had agreed to present a weakened case in court, and I was ruining the scheme.”
The plan was for Smith to deliver a weak argument in court, ensuring a victory in the landmark case for Eli Lilly. Smith and the plaintiffs would get their lavish payout, while Eli Lilly would score a huge PR win for Prozac.
Judge Potter was especially outraged because toward the trial's conclusion he'd asked both sides if they'd made a secret settlement, and both sides denied it. After lying to the judge, Smith and Eli Lilly and Co. then went on with the rigged trial. In retrospect, says Breggin, the aim of discrediting him as an expert witness for future Prozac suits was also a part of the overall settlement. The strategy failed only because Breggin had prepared his testimony despite Smith's obstruction and because he'd found critical documents Smith had purposely withheld from him.
Judge Potter was having none of Eli Lilly and Smith’s duplicity, and decided to change the verdict. He threw out the jury verdict and changed it to a settlement with prejudice by Eli Lilly and Co. Both Smith and Eli Lilly then appealed the judge's decision to the Kentucky Supreme Court. The Supreme Court quashed the appeal, concluding Eli Lilly had “manipulated” the judicial system and further opined that Eli Lilly might even have committed “fraud.”
Although the mass media covered the original fake victory by Eli Lilly and Co, only the British news agency Reuters made any mention of the reversal against the drug company. As a result of the media silence, the rigged trial was a massive legal, public relations, and political victory for Eli Lilly and Co.
Fixing jury trials is the kind of activity most people would associate with the mob. Big Pharma is indeed an organized crime ring, but it stays on the right side of the law by buying the favours of those in the regulatory, research and medical fields. Unlike mafia crime families, Big Pharma can overtly lobby Washington through organizations like the Pharmaceutical Research and Manufacturers of America (PhRMA), which spent $57 million on state and federal lobbying in 2016 alone.
Lying is Par for the Course Among Drug Company Manufacturers
Eli Lilly is hardly the only drug company that got ‘creative’ with its SSRI research data. Another instructive and well-documented example is the notorious Study 329. In July 2001, a team of GlaxoSmithKline-funded researchers published a comparison of GSK's SSRI Paxil (paroxetine) with the tricyclic antidepressant imipramine and also a placebo. The study recruited 275 adolescents aged 12-18 suffering a current episode of major depression of at least eight weeks’ duration.
The GSK-funded researchers concluded: “Paroxetine is generally well tolerated and effective for major depression in adolescents.” In the adverse events section of their paper, only 5 subjects experienced "suicidal ideation/gestures", of which 2 were hospitalized.
Study 329 was the biggest clinical trial of paroxetine on youngsters and the positive 2001 article was influential in supporting the use of antidepressants in adolescents.
But there was a wee problem:
Study 329 was a complete sham.
A mere two years later, in June 2003, the UK government warned physicians against prescribing paroxetine (which was marketed there under the name Seroxat) to children under 18.
Needless to say, that’s an unusual step to take against a drug that “is generally well tolerated and effective for major depression in adolescents.”
In reality, paroxetine is nothing of the sort.
In early 2007, the BBC investigative program Panorama aired a documentary on Paxil and Study 329. The documentary publicized hundreds of secret emails from GSK, uncovered during litigation, showing GSK knew years before publication of Study 329 that Paxil was not effective in treating adolescents for depression.
With regards to Study 329 specifically, a 5 March 2001 from a public relations executive working for GSK said: “Originally we had planned to do extensive media relations surrounding this study until we actually viewed the results. Essentially the study did not really show [Paxil] was effective in treating adolescent depression, which is not something we want to publicize.”
The lead author listed on the Study 329 paper was a Dr Martin Keller, then chief of psychiatry at Brown University, Rhode Island. Keller remains Professor Emeritus of Psychiatry and Human Behavior at Brown, despite a dubitable career that includes being investigated in 2009 by the US Senate Finance Committee, led by Senator Chuck Grassley, as one of a number of medical academics with a serious conflict of interest in his medical research.
Those conflict of interest allegations arose from the large sums of money Keller has received from drug companies. Keller has enjoyed millions of dollars of largesse from the pharmaceutical industry, much of it from companies whose drugs he was studying and touting at conferences and in medical journals. In 1998, for instance, the same year Keller published positive findings about Zoloft (sertraline) in the treatment of depression, Keller earned $218,000 in personal payments alone from Pfizer, the maker of Zoloft; he also received $77,400 from Bristol Myers Squibb, the maker of Serzone (nefazodone), the same year he was lead author on a positive study about the antidepressant. Serzone has since been taken off the market because it causes liver failure.
Between 2006 and 2010, Keller also received US $7 million in research grants from the National Institutes of Health (read: Taxpayers).
When asked during a 2006 US District Court deposition whether he had personally viewed the Study 329 data, Keller's answer was highly revealing:
“I've reviewed data analytic tables. I don't recall how raw it was, and I'm not trying to be facetious, but what I mean is that, you know, there are different levels of - how to put this - of organizing data that statisticians do. So, you know, the most primary level, the huge printouts that, you know, that list items by item number, you know, item numbers and variable numbers and don't even have words on them, I tend not to look at those. I -- I do better with words than I do with symbols.”
In other words, when presented with printouts of figures from the study, Keller admits he tends not to look at them because, you know, he’s not sure what they mean. He does better with words, you see.
In the end it didn't really matter, because the Study 329 paper was in fact written by a ghost-writer, a woman named Sally Laden at Scientific Therapeutics Information, in Springfield, New Jersey. A letter from STI to Keller told him all the necessary materials were enclosed so he could submit Study 329 for publication, even down to the covering letter, which came with the following instruction: "Please retype on your letterhead and revise if you wish."
Five days later, Keller thanked Laden for preparing the manuscript and providing instructions for submission:
“You did a superb job with this. Thank you very much. It is excellent. Enclosed are rather minor changes from me, Neal, and Mike (two of Keller’s listed co-authors)…”
 Sally K. Laden, the real author of the Study 329 manuscript.
Sally K. Laden, the real author of the Study 329 manuscript.
The first medical journal approached by GSK rejected the paper, so they tried another: The Journal of the American Academy of Child & Adolescent Psychiatry, whose then Editor, Dr Mina Dulcan, boasted to BBC: "We rank, and this is a worldwide ranking, we rank number one in child mental health and number two in paediatrics."
As the BBC discovered, some of the journal’s peer reviewers raised important concerns with the study:
"Overall results do not clearly indicate efficacy. Authors need to clearly note this," noted one.
Another pointed out: "A relatively high rate of serious adverse effects was not addressed in the discussion."
And further asked: "Given the high placebo response rate, are these drugs an acceptable first line therapy for depressed teenagers?"
Despite the concerns of its peer reviewers, the journal went ahead and published the study. When the BBC asked Dulcan if she had any regrets about publishing the study, she replied:
"Oh I don't have any regrets about publishing at all. It generated all sorts of useful discussion which is the purpose of a scholarly journal."
Dulcan seems to be confusing the purpose of a peer-reviewed journal with that of an Internet chat forum or a community discussion group. The purpose of a peer-reviewed “scholarly” journal is not to get people chin-wagging, but to help advance knowledge by presenting research that has been carefully scrutinized to ensure proper scientific protocols were met. To that end, the Journal of the American Academy of Child & Adolescent Psychiatry failed miserably.
Study 329 Re-Examined
In 2013, in response to rampant misreporting of outcomes of randomized controlled trials (RCTs), an international group of researchers called on funders and investigators of abandoned (unpublished) or misreported trials to publish undisclosed outcomes or correct misleading publications. This initiative was called “restoring invisible and abandoned trials” (RIAT) and, not surprisingly, Big Pharma wasn't exactly thrilled by the proposal.
The RIAT researchers identified Study 329 as an example of a misreported trial in need of reanalysis. And so on 14 June 2013, the RIAT researchers asked GSK whether it had any intention to reanalyse any of the trials it sponsored, including Study 329. GSK did not signal any intent to publish a corrected version of any of its trials. Despite the abundance of evidence to the contrary, GSK again insisted Study 329 “accurately reflects the honestly-held views of the clinical investigator authors” and did “not agree that the article is false, fraudulent or misleading.”
While insisting it had nothing to hide, GSK nonetheless made it difficult for the RIAT researchers to access to full study data. After negotiation, GSK made available all 275 case report forms for patients entered into Study 329. These forms, however, which totalled about 77,000 pages, were only made available through a remote desktop facility which, according to the researchers, "made it difficult and extremely time consuming to inspect the records properly."
When the RIAT researchers published the results of their Study 329 reanalysis in 2015, the results painted a very different picture to the rosy one presented in the original Keller et al paper.
Paroxetine (and imipramine) was no more effective than a placebo pill in treating adolescent depression.
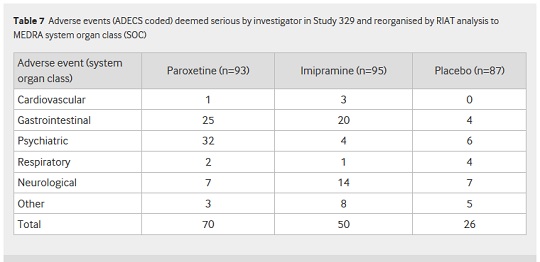
Paroxetine proved itself to be more than just useless, however. As you can see from Table 7 of the RIAT paper, published in BMJ on 16 September 2015, there was a far higher incidence of adverse effects in the paroxetine and imipramine groups compared to placebo (70, 50 and 26 adverse events, respectively):
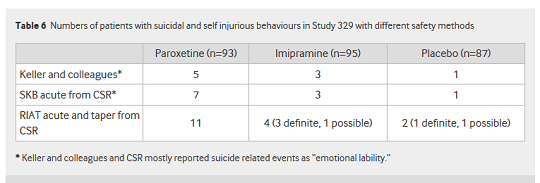
And as Table 6 from the paper shows, there was a major difference between the frequency of self-harm, suicidal thinking and suicide events reported for paroxetine by Keller et al, and that uncovered by the RIAT team:
The RIAT researchers uncovered eleven instances of suicide and self-injury in the paroxetine group – more than twice the number cited in the Study 329 paper!
Sertraline and Duloxetine: More Hiding of Data
Such appalling misrepresentation was in no way limited to Prozac and Paxil. Similar shiftiness was uncovered during a 2014 examination of 244 trials of six psychotropic drugs by Hughes et al.
Those six drugs included three anti-psychotic agents, an ADHD drug, the SSRI sertraline (Zoloft, by Pfizer) and the SSNRI duloxetine (Cymbalta). Hugh et al's reanalysis of the studies involving those drugs found:
“Most deaths (94/151, 62%) and suicides (8/15, 53%) reported in trial summaries were not reported in journal articles."[Bold emphasis added]
Read that again: In trials involving psychiatric drugs, including SSRIs and the newer SNRIs, most deaths and suicides occurring in those trials are not reported in the published papers.
That, it goes without saying, is disgraceful.
Let’s take a closer look at the results for sertraline and duloxetine. Of sixteen sertraline studies from the early 2000s, only 7 were published – and only 2 of those 7 studies reported on adverse events.
The total number of "suicidal ideation, attempts, injury" reported in those trials was 0.
However, the summaries for the 7 published trials retrieved from clinicalstudyresults.org (the former online public registry sponsored by PhRMA) in fact showed 5 instances of suicidal behaviour, and the summaries for the 9 unpublished studies showed a further 10.
Fifteen actual observed instances of suicidal behaviours, versus none reported in the published papers!
Deadly Duloxetine
Duloxetine is what is known as a selective serotonin–norepinephrine reuptake inhibitor (SSNRI). These are a newer class of drugs that supposedly increase the bioavailable amount of not just serotonin, but also norepinephrine (noradrenaline). Just like the SSRIs, no-one really knows the exact mechanisms of SSNRIs.
What we do know about SSNRIs is that, just like SSRIs, they are overrated, overprescribed, and dangerous.
Duloxetine was brought to market by none other than Eli Lilly in 2004, and is used to treat major depressive disorder, generalized anxiety disorder, fibromyalgia, neuropathic pain and stress urinary incontinence.
When Hughes et al examined the data from trials investigating duloxetine as a psychiatric intervention, they found 35 published trials that reported 33 suicidal events among those taking the drug. However, the summaries for these same trials retrieved from the now defunct clinicalstudyresults.org listed 40 suicidal events among the duloxetine subjects. Hughes et al discovered another 13 unpublished trials, for which the trial summaries reported a further 10 suicidal events among duloxetine subjects.
Drug Industry-Sponsored Research is a Farce
The Study 329 debacle highlights how every step of the clinical trial process can be corrupted by Big Pharma:
-Trial results are manipulated and even fabricated to make the studied drug appear safer and more effective than what it really is;
-Crucial adverse event findings such as elevated suicide risk are excluded from papers, before they are submitted for peer review;
-Ghost writers are hired to write scientific papers, which are then passed off as the creation of the lead researcher and his team. Often, these researchers do not fully scrutinize or even understand the data they are signing their names to.
-Public relations specialists - as opposed to detached, impartial scientists - determine how this ‘research’ will be promoted to the medical profession and media. This promotion typically emphasizes PR spin over fact.
-Medical journals, who receive much of their revenue from drug company advertising, proceed to publish this dubious research, even when peer reviewers raise serious concerns as to its quality and veracity.
-Instead of distancing themselves from researchers with questionable ethics, universities happily retain them due to the large amounts of drug company money and government research grants they attract to the university.
Given that such sloppiness and outright deceit seems to be routine procedure in research involving antidepressants, the obvious question is why should we trust drug companies, the researchers they fund, and the medical profession in general when they claim that SSRIs (and SSNRIs) do not raise suicide risk in adults?
The answer is simple:
We shouldn’t.
Randomized, placebo-controlled clinical trials were once considered the gold standard for testing the safety and efficacy of new drugs. When conducted in an impartial and exacting manner, such trials are fully deserving of that honour. Unfortunately, drug company money has so thoroughly polluted the field of clinical research, it’s now almost impossible to accept at face value the results of any drug company-sponsored trial.
We shouldn’t have to rely on investigative journalists and uncorrupted independent researchers to reanalyse studies in order to finally bring us the true results of drug trials – but, sadly, that is now the case.
Deadly Prozac
It speaks volumes when you have to rig a jury trial to cover up the adverse effects of your star anti-depressant drug. Like all the SSRIs, Prozac is highly overrated and dangerous garbage.
And this was known a long time ago.
The first published case series linking Prozac to suicidal behaviour was published way back in 1990, by Dr Martin Teicher, a psychiatrist at McLean Hospital and Harvard Medical School, and his colleagues Dr Jonathon Cole and Carol Glod. They reported on six depressed patients, aged 19 to 62. All had been free of recent serious suicidal ideation, but developed intense, violent suicidal preoccupation after 2-7 weeks of fluoxetine treatment. The suicidal thoughts persisted for as little as 3 days and as long as 3 months after discontinuation of fluoxetine.
After publication of the Teicher paper, more case reports began appearing in the literature. Among these was a 1991 report by a team of Yale doctors titled “Emergence of Self-Destructive Phenomena in Children and Adolescents during Fluoxetine Treatment.” Six of the 42 (14%) youngsters in their cohort became aggressive and/or violent, self-injurious and suicidally ideative while taking fluoxetine. The authors gave brief case descriptions of each subject, and each case makes for disturbing reading. The description of "F", a 12-year-old boy who participated in a double-blind, placebo-controlled fluoxetine protocol for treatment of OCD, was especially striking given what subsequently occurred at Columbine. Thirty-eight days after beginning the protocol, F experienced a violent nightmare about killing his classmates until he himself was shot. He awakened from it only with difficulty, and the dream continued to feel "very real".
He reported having had several days of increasingly vivid "bad dreams" before this episode; these included images of killing himself and his parents dying. When he was seen later that day he was agitated and anxious, refused to go to school, and reported marked suicidal ideation that made him feel unsafe at home as well.
F was hospitalized twice. He gradually improved, but then three weeks after his last hospitalization, he was placed back on fluoxetine. He became acutely suicidal until the fluoxetine was stopped a second time.
SSRIs Increase Suicide Risk Across All Age Groups
In 2005, BMJ published a systematic review of suicide risk reported in randomised controlled trials (RCTs) of SSRI drugs. The review was headed by Dean Fergusson, a scientist at the Ottawa Health Research Institute in Ontario, Canada. To warrant inclusion in the review, the trials had to compare a SSRI with either placebo or an active non-SSRI control. Some 702 trials including 87,650 patients met the inclusion criteria. However, only 345 of these 702 trials, representing 36,445 patients, reported the number of suicide attempts and were thus included in the analysis. The researchers found larger trials tended not to report suicide attempts.
When SSRIs and tricyclic antidepressants were compared, similar numbers of non-fatal and fatal suicides were reported (0.56% vs 0.65%). In trials comparing SSRI drugs with other active treatments (most commonly the MAO inhibitor moclobemide and psychotherapy), the rates were 0.65% and 0.43%, respectively.
As for SSRIs versus placebo, there were initially 50,570 subjects in the trials comparing SSRIs with placebo, but suicide data was only reported for 18,413 of them. Among this greatly reduced pool of subjects, non-fatal and fatal suicides were 2.28 more likely among those receiving SSRIs compared with placebo. Twenty-seven of the 10,557 SSRI patients were reported to have attempted or completed suicide, compared to 9 of the 7,856 placebo patients (0.26% versus 0.11%, respectively). Four of the SSRI suicides were fatal, compared to 3 in the placebo group. The increased risk was seen across all age groups except for over 60 years, where there was one reported suicide attempt in both the SSRI and placebo groups.
Most of this increased risk came from non-fatal attempts; there was no significant difference in the risk of fatal suicide attempts.
However, as we now know, drug companies routinely lie and conveniently omit data about the suicide rate of their SSRI trial participants. Meaning that the increased suicide risk imparted by SSRIs was very likely much greater than that revealed in the data Fergusson et al had access to.
Evidently, Fergusson et al harboured similar suspicions, so they took an additional step. They contacted investigators from a random sample of 35 (10%) trials that did not report suicide attempts. Of the 17 responders, two reported suicide attempts, seven reported no suicide attempts, and eight confirmed that these data were not collected.
Of the two responses that reported suicide attempts, one reported a non-fatal suicide attempt in the SSRI group and none in the placebo group, and the other reported a non-fatal suicide attempt in the SSRI group and two non-fatal attempts in the group taking tricyclic antidepressants.
Heaven knows what the true magnitude of the increased suicide risk imparted by SSRIs really was. As Fergusson et al surmised: "A more accurate estimation of risks of suicide could be garnered from investigators fully disclosing all events."
Here’s what we do know: Even when using biased and incompletely reported data from drug company trials, where subjects were carefully screened prior to entry and most investigators neglected to report suicidal behaviour, SSRIs still imparted a 2.3 times greater risk of suicide attempts.
More Recent Research Confirms these Results
Researchers from the Nordic Cochrane Centre in Copenhagen, Denmark set out to quantify the risk of suicidality and violence when SSRI and SSNRI drugs are given to healthy adult volunteers with no signs of a mental disorder.
After screening almost 6,000 publications, they found 130 trials that fulfilled their inclusion criteria. The trials were generally uninformative; 97 trials did not report the randomisation method, 75 trials did not report any discontinuations and 63 trials did not report on adverse events.
They found thirteen trials that reported on the adverse events under investigation, but 2 of these were crossover trials that didn't report the data for each period separately (a crossover trial is one in which each volunteer receives both the treatment and placebo, in separate periods).
And so only eleven of the 130 published trials contained the data required by the researchers for their meta-analysis.
Which again underscores a point I made in Part 2: Overall, the quality of clinical research involving antidepressant drugs is terrible.
In this instance, there was simply no excuse for such crappy and poorly-reported research; the median publication year of these trials was 2008 ... not 1908.
The drugs investigated in these trials were citalopram, escitalopram, fluoxetine, paroxetine, sertraline and venlafaxine. The median age of participants was 30 years; 40% of the volunteers were women.
Based on the data they had available, treatment of adult healthy volunteers with antidepressants almost doubled their risk of suicide- and violence-related harm (odds ratio 1.85). The number needed to treat to harm one healthy person was 16.
As the researchers noted, “There can be little doubt that we under-estimated the harms of antidepressants, as we only had access to the published articles for 11 of our 13 trials.”
In one of the two crossover trials the researchers excluded, a healthy volunteer committed suicide. She had received duloxetine in increasing doses for 16 days, tapered off the maximum dose of 400 mg daily in just four days, and killed herself four days later while on placebo. The authors, "several of whom were employees of Eli Lilly or owned stock in the company," judged her suicide "to be unrelated to study drug treatment", even though it is well known suicide risk is high when an antidepressant is stopped abruptly.
The Danish researchers asked Eli Lilly for access to anonymised data for the volunteer who committed suicide, as they wanted to know how it could possibly state the suicide was unrelated to duloxetine, but the company refused.
In another of Eli Lilly’s studies, a healthy 19-year-old student who had taken duloxetine in order to help pay her college tuition hanged herself in a laboratory run by Lilly. It turned out that missing in the FDA’s files was any record of the college student and at least four other volunteers known to have committed suicide, and Lilly admitted that it had never made public at least two of those deaths.
Antidepressants Raise the Risk of Suicidal Behaviour in Adults
It’s time for drug companies, the medical profession and regulatory agencies like the FDA to stop lying through their teeth about antidepressant drugs. SSRI and SSNRI drugs unquestionably raise the risk of suicidal behaviour, not just in adolescents, but also in adults. By just how much is hard to discern, because drug companies are staggeringly dishonest entities who are happy to suppress suicide-related data. They do this so they can go ahead and get approval for and then aggressively market drugs they know full well will cause people to kill themselves.
Based on the watered down data that Fergusson and the Danish researchers were forced to work with, it would seem antidepressants at least double the risk that users will engage in suicidal behaviours.
But the research of Hughes et al, which uncovered fifteen instances of suicidal behaviours in sertraline trials versus the 0 reported by Pfizer, suggests the real risk is much, much higher.
Does This Stuff Even Work?
One doctor I know tried to brush off the increased suicide risk of SSRI treatment by claiming it was unfortunate, but a small price to pay in light of all the people these drugs are [allegedly] helping.
In other words, folks who become suicidal after being prescribed SSRIs are simply taking one for the team.
I find this reasoning abhorrent, for two reasons:
-Subjecting unsuspecting individuals to an increased suicide risk for the ‘greater good’ is not ethical and not acceptable - especially when there are far less damaging treatments available;
-There is little-to-no evidence SSRI and SSNRI antidepressants actually work, beyond their effect as active placebos.
That last statement is guaranteed to get a whole lot of vested panties in a twist, so stay tuned ... in due course I'll be releasing a project that discusses the evidence for this in detail.
I’ll also discuss therapies for depression and anxiety that clinical research has shown to be effective, without the plethora of harmful side effects caused by antidepressants.
Hasta luego,
Anthony.
[Article updated 16 Aug 2019].
---
Anthony Colpo is an independent researcher, physical conditioning specialist, and author of the groundbreaking books The Fat Loss Bible, The Great Cholesterol Con and Whole Grains, Empty Promises.
For more information on Anthony's books, click here.
---
The Mandatory “I Ain’t Your Mama, So Think For Yourself and Take Responsibility for Your Own Actions” Disclaimer: All content on this web site is provided for information and education purposes only. Individuals wishing to make changes to their dietary, lifestyle, exercise or medication regimens should do so in conjunction with a competent, knowledgeable and empathetic medical professional. Anyone who chooses to apply the information on this web site does so of their own volition and their own risk. The owner and contributors to this site accept no responsibility or liability whatsoever for any harm, real or imagined, from the use or dissemination of information contained on this site. If these conditions are not agreeable to the reader, he/she is advised to leave this site immediately.