In the West, three drugs currently dominate the COVID-19 vaccine limelight: The Pfizer-BioNTech and Moderna mRNA drugs, and the Oxford-AstraZeneca COVID-19 vaccine. The latter, as the name indicates, is the result of a collaboration between Oxford University and multinational drug giant AstraZeneca.
In my last post, I discussed the shady science behind the Pfizer-BioNTech drug, and how the new vaccine is increasingly associated with a most untoward side effect: Death.
As of this writing, the Pfizer-BioNTech and Moderna drugs are the only 'vaccines' that have been granted "emergency use" authorization in the US. Between the two of them, they've racked up 288 deaths on the VAERS reporting system from their December rollout through to 21 January 2021 in the US and its territories.
Both of these dubious drugs have also enjoyed "emergency use" approval in the UK, along with the Oxford-AstraZeneca vaccine. As of 24 January 2021, there have been 22,820 adverse events reported for COVID-19 vaccines on the UK's MHRA Yellow Card reporting system; 16,756 of these were for the Pfizer-BioNTech vaccine, 6,014 for the Oxford-AstraZeneca vaccine, and 50 for unspecified brand/s.
When it comes to deaths, during the same period the MHRA received 107 reports for the Pfizer/BioNTech vaccine in which the patient died shortly after vaccination, 34 reports for the Oxford-AstraZeneca vaccine and 2 where the brand was unspecified.
That's quite a feat, considering the first approved COVID vaccine jab in the UK was administered only 20 days earlier, on 4 January 2021.
But while the UK authorities seem happy to label just about anybody that passes away as a COVID-19 victim, they are extremely reluctant to acknowledge the dangers associated with the poorly-tested COVID vaccines. Lung cancer victims, people with blood clots, and children with Kawasaki disease are among the unlikely patients who have been misdiagnosed with COVID in the UK. And when elderly people with other pre-existing conditions pass away after testing positive for COVID, the coronavirus - and not their far more serious co-morbidities - is listed as the cause of death.
But when it comes to holding vaccines accountable for the damage they appear to be causing, the UK Government and MHRA suddenly becomes extremely reluctant to upset its very close friends at Big Pharma. And harbor no illusions: If the MHRA and pharmaceutical industry were any closer, they'd be joined at the hip. Since changes made by Margaret Thatcher in 1989, the MHRA has relied on the pharmaceutical industry for 100% of its funding.
And just like the US FDA and other regulatory agencies around the world, the supposedly independent agency is plagued by the infamous "revolving door" phenomenon, in which staff routinely shift between employment at the agency and drug companies.
Here's but a mere sampling of the links between the drug industry and UK health authorities, who are supposed to be acting in the public interest:
Sir Patrick Vallance is the Government Chief Scientific Adviser and also a key member of the Scientific Advisory Group for Emergencies (SAGE) which is steering the UK's dystopian response to COVID-19.
From 2006 to March 2018, Vallance worked for the corrupt GlaxoSmithKline. He began as head of drug discovery, and in 2012 was appointed head of research and development at GSK. He went directly from GSK to his post as the UK Government chief scientific adviser. Several months later, it emerged that Vallance owned 43,111 shares worth £600,000 (US $824,000) in GlaxoSmithKline, which is currently developing a COVID vaccine.
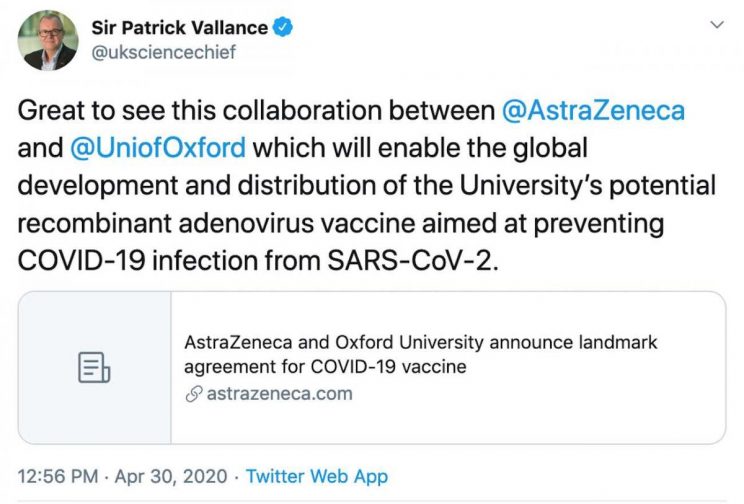
 Patrick Vallance, UK Government Chief Scientific Adviser, former head of research and development at GlaxoSmithKline, and proud owner of over $800,000 worth of GSK shares.
Patrick Vallance, UK Government Chief Scientific Adviser, former head of research and development at GlaxoSmithKline, and proud owner of over $800,000 worth of GSK shares.
Ian Hudson, MHRA chief executive from 2013 to 2019, is a former vice president of GSK (then SmithKline Beecham). In 2019, Hudson became Senior Advisor, Integrated Development, Global Health for the intensely pro-vaccine Bill and Melinda Gates Foundation.
Former MHRA Director of Inspection, Enforcement and Standards, Gerald Heddell, worked at UK's National Health Service until 1978, then left to enjoy senior roles at The Wellcome Foundation, Glaxo Wellcome and GlaxoSmithKline. He then returned to bureaucratic life in 2005 with a senior posting at the MHRA.
Prior to joining the MHRA in 2008, Senior Medical Assessor Dr Nithyanandan Nagercoil, worked for a generic pharmaceutical company and then a global contract research organization performing clinical trials on behalf of drug companies. During his time as a clinical researcher, he helped conduct trials for AstraZeneca and Pfizer, among others.
MHRA documentation shows current MHRA Non-Executive Directors Anne-Toni Rodgers and Amanda Calvert are shareholders in AstraZeneca. The current Chair of the MHRA Board, Stephen Lightfoot, is a shareholder in GlaxoSmithKline, while MHRA Non-Executive Director Professor Liam Smeeth is the beneficiary of research grants from GlaxoSmithKline.
Little surprise then, that instead of urging the public to be cautious of COVID vaccines, the MHRA bends over backwards to portray them as safe and to dismiss their reported adverse events as unrelated coincidences.
Through to 24 January 2021, the overall UK reporting rate of adverse events for the Oxford-AstraZeneca and Pfizer-BioNTech vaccines was around 3 Yellow Cards per 1,000 doses administered. The MHRA eagerly cites this seemingly low incidence as proof the vaccines are safe and that their benefits outweigh the risks. This is a most disingenuous line of argument considering the well-established fact that reporting systems like VAERS and Yellow Card are notoriously prone to under-reporting. They are voluntary "passive surveillance" systems and rely on awareness and motivation to report. Unlike the massive propaganda campaigns urging social distancing, mask-wearing and ratting on your neighbours, there are no prime time TV ads urging people to report vaccine side effects. And while medical professionals have a higher awareness of these systems than the public, many still don't bother to report drug side effects because of the time and paperwork involved.
Systems like VAERS and Yellow Card, therefore, capture only a small percentage of vaccine adverse events. An illuminating example is the well-established vaccine side effect of anaphylaxis, which can be life-threatening. A recent CDC study found VAERS sensitivity for capturing anaphylaxis after seven different vaccines ranged from 13% to 76% of actual cases. The VAERS system captured only 13% of cases after influenza vaccine, 21% of cases after varicella vaccine, 24% of cases after both the live attenuated zoster and quadrivalent human papillomavirus vaccines, 25% of cases after the combined measles, mumps and rubella (MMR) vaccine, and 76% of cases after the 2009 H1N1 inactivated pandemic influenza vaccine.
These figures indicate the true rate of anaphylaxis events from vaccines could be as much as 8 times higher than the VAERS figures.
Anaphylaxis is not the only condition greatly underrported on the government databases. For Guillain-Barré syndrome (GBS), a nasty autoimmune disorder, VAERS captured only 12% of cases after the 2012-13 inactivated seasonal influenza vaccine and 15-55% of cases after the 2009 H1N1 vaccine.
But the MHRA doesn't give a hoot. It claims "the majority of [Yellow Card vaccine adverse event] reports were in elderly people or people with underlying illness."
Right, just like COVID-19 - which is being portrayed as humankind's Armageddon event.
When it comes to deaths, the pharma-affiliated MHRA says: "Review of individual reports and patterns of reporting does not suggest the vaccine played a role..." And of the tens of thousands of adverse event reports that have accumulated in less than a month, the MHRA is similarly unmoved, claiming, "There are currently no indications of specific patterns or rates of reporting that would suggest the vaccine has played a role."
Yeah, no worries. By the way, how's the price of your AstraZeneca and GSK shares travelling, dear SAGE and MHRA staff?
The First Non-mRNA Big-Name COVID Vaccine
Unlike the Moderna and Pfizer-BioNTech mRNA 'vaccines', the Oxford-AstraZeneca vaccine is based on a chimpanzee adenovirus which causes colds in our closest genetic cousins. Oxford researchers took this adenovirus and genetically altered it to contain the SARS-CoV-2 spike protein gene. The chimp adenovirus, we are told, is replication deficient, meaning it cannot cause further infection and simply acts as a vector for the COVID S-protein gene.
So does that mean it's all fine and dandy to let some smiling nurse prick you in the arm and inject you with the Oxford-AstraZeneca vaccine?
Only if you like receiving poorly-studied drugs manufactured by criminals.
The Terribly Inadequate Research Behind the the Oxford-AstraZeneca COVID-19 Vaccine
The first peer-reviewed paper proclaiming the alleged efficacy and safety of the Oxford-AstraZeneca COVID-19 vaccine (also known as the "ChAdOx1 nCoV-19 vaccine" or "AZD1222") was published online in the Lancet on 8 December 2020. This was the data that supposedly showed the vaccine was Safe and Effective!™ and justified its approval by UK authorities on 30 December 2020
After reading through the paper and the supplementary material for this study, and considering the nefarious background of AstraZeneca, my appraisal of this study is not a kind one. To be frank, I don't think the study is worth the paper it's written on. It's a mess of woefully incomplete, inadequate and often irrelevant slop, brought to us by yet another criminally reckless pharma multinational sporting a long and shameful corporate rap sheet.
Anyone who claims the vaccine has been proven safe and effective is either a shameless liar or, as they say in England, "bloody bonkers!"
Let me count the ways in which the research behind the Oxford-AstraZeneca vaccine is a bad joke.
1. The Lancet Paper Used Part of the Data from Part of a Study that is Still Ongoing
As with the Pfizer-BioNTech and Moderna vaccines, the research supposedly supporting the Oxford-AstraZeneca vaccine is nowhere near complete. There are four trials in progress of the Oxford-AstraZeneca vaccine, and according to Clinicaltrials.gov there are numerous more yet to kick off. It is the four trials in progress that are mentioned in the Lancet paper. Of those four trials, 23,848 participants were enrolled in Phase 2 and 3 clinical trials in the UK, Brazil, and South Africa between 23 April and 4 November 2020.
But only half of those subjects were included in the interim analysis that appeared in the December Lancet article. Only three of those four trials, and only 11,636 of the participants were included in the Lancet analysis (7,548 subjects in the two UK trials, 4,088 in Brazil).
2. Not Only is the Phase 3 Testing Incomplete, they Haven't Even Finished the Phase 2 Trials!
Under normal circumstances - in which governments and billionaire-owned media outlets have not colluded to portray a flu virus with a <0.5% infection fatality rate as the deadliest 'emergency' ever faced by mankind - successful completion of Phase 3 clinical trials is a prerequisite to getting a vaccine or any other drug approved. The Phase 3 research, of course, is preceded by smaller Phase 1 and 2 clinical trials which examine tolerable dosages and side effects of the new drug. The information gleaned from these smaller trials is then used to design Phase 3 trials. But the Lancet paper is an interim analysis featuring not only incomplete Phase 3 research, but incomplete Phase 2 research!
One of the UK trials is listed as a Phase 2/3 endeavour, while the Brazil trial is designated Phase 3. The other UK trial and the South African study are listed as Phase 1/2 studies.
Again, anyone who tells you the current batch of vaccines has been thoroughly tested and proven Safe and Effective!™ is so full of caca you can probably smell them from Jupiter. The truth is that the whole COVID vaccine charade is a massive rush job.
3. The Oxford-AstraZeneca COVID-19 Vaccine Trials are Not Double-Blind
Phase 3 drug trials are supposed to utilize the double-blind approach to prevent subjects and researchers alike from knowing who is in what group. If the subjects know what treatment they are getting, then the well-known placebo effect could sway their perceptions and behaviour during the trial, and hence influence the results. And if the researchers know who is getting the treatment, then their beliefs about that treatment or their financial ties to the developers of that treatment could also lead to biased results.
But guess what? Only one of the four trials (South Africa) was conducted under double-blind conditions - and that was the one not included in the interim analysis. The rest of the trials were "participant blind" only, meaning that the subjects were not told what treatment they were getting.
While the participants were not told which group they were in, almost all the researchers knew. The only exception was a "central, blinded adjudication committee" that assessed the data of participants testing positive for SARS-CoV-2 during the trials. Each case was assessed by the adjudication committee and classified according to the WHO severity grading scale for COVID-19 (0 = uninfected, 10 = dead). These adjudicated results were used for the pooled analyses that feature in the Lancet paper.
4. Conflict of Interest is Rampant Among the Trial Researchers
The information fed to this allegedly blinded committee, of course, came via an army of non-blinded researchers, many of whom have a financial interest in the vaccine being studied. From the authors listed in the main Lancet paper, three are employees of AstraZeneca; the supplementary material lists another eight researchers directly employed by AstraZeneca. Another researcher listed in the main paper reported receiving grants and personal fees from AstraZeneca, while another reported receiving "non-financial" support from the drug giant.
AstraZeneca also funded the study, along with several other outfits including the Bill & Melinda Gates Foundation and the Coalition for Epidemic Preparedness Innovations (CEPI), which held a pandemic simulation in October 2019 in New York which magically predicted the coronavirus outbreak that followed shortly after, almost down to a tee.
AstraZeneca entered into a partnership with Oxford University to further develop the ChAdOx1 nCoV-19 vaccine, which means a massive payday not just for AstraZeneca, but Oxford University. Oxford clearly has a huge vested interest in the vaccine, and the supplementary data lists a hefty 106 researchers belonging to the Oxford Vaccine Group.
A company called Vaccitech was also an early collaborator in the development of the vaccine. According to the disclosure section at the end of the Lancet paper, one of the researchers is a co-founder of Vaccitech and named as an "inventor on a patent covering use of ChAdOx1-vectored vaccines and a patent application covering the AZ-Oxford vaccine (PCT/GB2012/000467)." Another is named as an inventor on a patent application covering the AZ-Oxford vaccine and is a consultant to Vaccitech for an unrelated project during the study. Another researcher is listed as a consultant to Vaccitech during the conduct of the study.
And yet another researcher reported "personal fees from Vaccitech" and "has a patent on ChAdOx1 licensed to Vaccitech (PCT/GB2012/000467), and might benefit from royalty income to the University of Oxford from sales of this vaccine by AstraZeneca and sublicensees."
So to reiterate, this was a study in which the overwhelming majority of researchers were non-blinded, and many of those researchers had a vested interest in the success of the vaccine they were studying and reporting on.
That does not comfort me at all.
Neither does the fact that supposedly respected universities like Oxford are more than happy to partner up with dubious outfits like AstraZeneca.
5. AstraZeneca is a Serial-Offending Corporate Criminal
The London-based AstraZeneca is the result of a 1999 merger of Britain’s Zeneca with Sweden’s Astra AB. The newly-merged drug giant didn't waste any time establishing its criminal palmares - in 2003 US federal officials announced AstraZeneca had pleaded guilty to criminal and civil charges relating to the illegal marketing of the prostate cancer drug Zoladex. The company agreed to pay $355 million, consisting of $64 million in criminal fines, a $266 million settlement of civil False Claims Act charges, and a $25 million settlement of fraud charges relating to state Medicaid programs.
In 2005 the European Commission fined AstraZeneca 60 million euros for misusing patent rules to shield its ulcer drug Losec (Prilosec in the US) from generic competitors. The penalty which was upheld by the European Court of Justice in 2012.
In 2007, a federal judge ruled AstraZeneca and two other companies had to pay damages in connection with overcharging Medicare and private insurance companies. The judge singled out AstraZeneca for acting “unfairly and deceptively” in its pricing of prostate cancer drug Zoladex. AstraZeneca was later hit with a $12.9 million judgment.
In 2009 AstraZeneca was one of four drug companies that entered into a settlement agreement under which they agreed to pay a total of $124 million to settle charges that they violated the federal False Claims Act by failing to provide required rebates to state Medicaid programs. AstraZeneca’s share of the total settlement amount was $2.6 million.
In 2010 AstraZeneca agreed to pay $103 million to settle a national lawsuit accusing the company of overcharging for Zoladex and Pulmicort Respules asthma medication.
That same year, the US Justice Department announced AstraZeneca would pay $520 million to resolve allegations it illegally marketed its anti-psychotic drug Seroquel for uses not approved as safe and effective by the FDA. Under the terms of the settlement, $302 million of the total was to go to the federal government and $218 million to state Medicaid programs. Among other things, the company was accused of having paid doctors to give speeches and publish articles (ghostwritten by the company) promoting those unapproved uses.
In 2011 AstraZeneca settled a related Seroquel case brought by state governments by agreeing to pay another $69 million.
In 2016, AstraZeneca was forced to fork out $5.5 million to settle a bribery probe by the SEC involving allegations staff used gifts and cash to persuade doctors to buy its drugs. The SEC alleged that the company's staff in Russia and China provided gifts, conference support, travel, cash and other inducements to state-employed healthcare providers to buy or prescribe the company’s products. The company also falsely recorded the improper payments in China and Russia as bonafide business expenses, the SEC said.
This is only a partial sampling of AstraZeneca's bad behaviour; for more, click here.
Needless to say, AstraZeneca spends a lot of time and money settling dishonesty and fraud cases. Is that really the kind of outfit you want to trust with your health?
6. Most of the Oxford-AstraZeneca COVID Vaccine Trials Did Not Contain a Non-Vaccine Placebo - and the One that Did Was Omitted From the Lancet Analysis
Yep, as if all the above isn't bad enough, there's another critical tidbit the pro-vaccine shills have failed to disclose when publicly pimping the AstraZeneca vaccine: In these trials, the overwhelming majority of 'control' subjects received, not an inert placebo, but a meningococcal vaccine. In other words, most of the Oxford-AstraZeneca vaccine trials are not vaccine versus no-vaccine trials, they are COVID vaccine versus meningococcal vaccine trials.
In the UK trial, control participants received two doses of the meningococcal vaccine. In the Brazilian trial, the control subjects received the meningococcal vaccine for their first shot, and a saline injection for their second. The only trial that used an inert (saline) placebo for both control shots was the South African study, but again, that was not included in the interim analysis.
So the data used to declare the Oxford-AstraZeneca vaccine Safe and Effective!™ did not even involve a comparison of people receiving the vaccine with those receiving no vaccine - it involved three vaccine versus vaccine trials.
The researchers' excuse for doing this in the UK trial was "to minimise the chance of accidental participant unmasking due to local or systemic reactions to the vaccine" (no rationale was provided for giving the meningococcal vaccine to Brazilian participants).
What they are claiming here is that they used the meningococcal vaccine in the UK trials as an "active placebo," to try and prevent the subjects from guessing they were in the COVID vaccine group by way of experiencing side effects. Given that the biggest danger of bias in this study came from the researchers, I'm not at all convinced. I suspect the real reason, one that they unwittingly allude to in their rationale for using the meningococcal vaccine, is that they knew a saline solution would produce less "local or systemic" side effects than the COVID vaccine. Using another vaccine as the control treatment would help even out the rate of adverse events and make the Oxford-AstraZeneca vaccine appear safer than what it really is.
7. The Participants Were Heavily Filtered During Recruitment and Do Not Represent the 'Real World'
In my previous post, I wrote how the only trial for the Pfizer-BioNTech vaccine involved an unusually healthy participant sample. Despite this, the vaccine is eagerly being injected into all and sundry, including frail elderly folks.
It's a similar story with the Oxford-AstraZeneca vaccine. One of the UK trials (code-named COV001) involved healthy adult participants who were enrolled only after screening to exclude those with preexisting health conditions. The other UK trial (COV002) targeted people working in professions with "high possible exposure to SARS-CoV-2, such as health and social care settings." The Brazilian trial included individuals with "stable pre-existing health conditions."
The average BMI of participants in the UK (and Brazil) trials was a hair over 25, compared to the actual UK average of 27.6.
None of the UK cohorts had a diabetes incidence higher than 2.5%, a stark contrast to the 2015 diabetes prevalence of 9% among UK adults.
None of the UK (or Brazil) cohorts had a respiratory disease incidence higher than 13%, even though some 20% of people in the UK have a history of asthma, COPD or another longstanding respiratory illness.
Based on these figures alone, we can see the study population featured in the Lancet analysis was not an ideal proxy for the general population. But the above discrepancies pale in comparison to the most glaring omission of all:
Older folks.
As with the Pfizer-BioNTech trial, you had to be 18 years or older to be eligible for participation in the AstraZeneca trials. But if you were over 55, it seems Oxford and AstraZeneca didn't want to know you, either.
Of the 2,741 participants in the UK COV001 trial, the number of those aged 56 and over was a big fat zero.
In the UK CV002 trial involving at risk workers, only 21% were aged 56 and over.
And in the Brazil (CV003) trial, only 10% of participants were aged 56 and older.
The trials featured in the Lancet analysis, in other words, are ill-equipped to inform us of the safety and efficacy of the virus in those 56 and over. And yet the vaccine is being urged with reckless abandon to people of all ages. Heck, the first person to receive the vaccine in the UK was an 82 year-old chap by the name of Brian Pinker. Not only that, but Brian was a kidney dialysis patient.
Just brilliant.
Boris Johnson, who appeared on TV emphatically declaring the COVID vaccine rollout was "safe" (free tip: Never take health advice from a pudgy politician who can't even comb his hair), was forced to defend the AstraZeneca vaccine after Germany declared it will not recommend the jab for over-65s. A draft recommendation from Germany’s vaccination advisory committee recently stated there was “insufficient data” on its efficacy for those aged 65 and over.
Johnson said he was not concerned about the decision and argued the vaccine provided “a good immune response across all age groups ... I think the MHRA, our own authorities, have made it very clear that they think the Oxford/AstraZeneca vaccine is very good and efficacious, gives a high degree of protection after just one dose and even more after two doses."
Yeah, but your authorities are in the pocket of Big Pharma, Boris.
Another country that remains unconvinced by Boris' bollockery is Spain, which recently banned the Oxford-AstraZeneca vaccine in over 55s. France, Italy, Austria and Norway have also imposed age limits on the Oxford-AstraZeneca vaccine.
 Dr Boris, staring at a vaccine vial.
Dr Boris, staring at a vaccine vial.
So the Research is Junk, But What Did it Find?
The total number of COVID-19 cases among those receiving the Oxford-AstraZeneca vaccine was 30 (0.5%) compared to 101 (1.7%) in the meningococcal vaccine group.
From 21 days after the first dose, there were ten cases hospitalised for COVID-19, all in the meningococcal vaccine group. Two of these were classified as severe COVID-19 by the researchers, including one death.
There were four non-COVID-19 deaths reported (one Oxford-AstraZeneca subject and three meningococcal vaccine subjects) that were all considered unrelated to the vaccines. The causes of death were determined to be from road traffic accident, blunt force trauma, homicide, and fungal pneumonia.
After a median 3.4 months of safety follow-up, the researchers claim only 84 events in the Oxford-AstraZeneca cohort and 91 in the meningococcal vaccine cohort.
The Pfizer-BioNTech study I discussed in my previous post recorded no COVID-related deaths among the 37,706 trial participants analyzed. The Oxford-AstraZeneca study has recorded a single allegedly COVID-related death among the incomplete sample of 11,636 participants featured in the Lancet paper. Needless to say, this is a statistically insignificant result that tells us next-to-nothing about the alleged life-saving properties of the Oxford-AstraZeneca vaccine. In fact, as it currently stands, there is not a shred of evidence to show either the Pfizer-BioNTech or Oxford-AstraZeneca vaccines can save even a single life when compared to no vaccine treatment.
Based on emerging clinical trial evidence conducted by non-criminal entities, I'm predicting administration of cheap and truly safe vitamin D supplements will blow the highly dubious COVID vaccines out of the water.
But Wait, There's More BS
Despite all this, a few days ago, a press release loudly declared "COVID-19 Vaccine AstraZeneca confirms 100% protection against severe disease, hospitalisation and death in the primary analysis of Phase III trials."
Wow! Complete, absolute, 100% protection from severe COVID disease, hospitalization and death! It sounds too good to be true.
And it is.
The entity that issued the press release was AstraZeneca which, as we've seen, is not exactly a beacon of ethics and honesty.
And the new primary analysis the release gushed about was a Lancet preprint, yet to be peer-reviewed, which can be found here.
Again, it involves only a portion of the Oxford-AstraZeneca trial population, this time solely from the UK COV002 trial. As you read through it, you'll be subjected to a truckload of gobbledegook about the "new UK variant" of COVID-19 called B.1.1.7. But when you scroll past all the waffle about B.1.1.7. and non-B.1.1.7 lineages and look at the actual number of cases, a far less newsworthy picture emerges.
There were a total of 250 "Primary Symptomatic" COVID-19 cases among the participants; 52 of 4,236 (1.2%) vaccine subjects, and 198 of 4,270 meningococcal vaccine subjects. This equates to incidence rates for symptomatic infection of 1.2% and 4.6%, respectively.
Those figures wouldn't be at all difficult to manipulate in a trial where clinical researchers rife with vested interests know who is getting the vaccine, and who isn't.
When it came to "Asymptomatic/Unknown infections," there were 96 of 4,236 vaccine subjects, and 112 of 4,270 meningococcal vaccine subjects. This gives incidence rates for asymptomatic/unknown infection of 2.3% and 2.6%, respectively.
Excuse me while I yawn.
And deaths?
There is no mention of mortality, or hospitalizations, or even severe illness, for that matter! Which means the AstraZeneca press release headline declaring this study "confirms 100% protection against severe disease, hospitalisation and death in the primary analysis of Phase III trials" is simply another example of the egregious bullshit for which drug companies have become notorious.
The Bottom Line
The fanfare that has been whipped up for the Oxford-AstraZeneca COVID-19 vaccine is based on garbage science that should be used in university courses - for teaching students how not to conduct clinical research. The company responsible for producing and marketing the vaccine, AstraZeneca, is dodgey as all get out, having racked up well over a billion dollars in fines and settlements for dishonesty offences. And to top it off, the researchers and health authorities responsible for the vaccine's research and its accompanying hyperbole have more conflicts of interest than a millipede has legs.
All this, it must be remembered, is to fight an absurdly over-hyped influenza virus that repeated studies show has an infection fatality rate of 0.5% or less. There is a significantly greater risk of dying from cancer, heart disease, car accident, a fall, or suicide. But I don't see any of these dystopian technocrats bringing the world to a grinding halt to prevent deaths from any of those causes. To the contrary, deaths from cardiovascular causes and suicide have risen as a result of the COVID stupidity and its associated lockdowns.
Think about that.
If You Found This Article Helpful, Please Consider Leaving a Tip
This site is self-funded and relies on reader generosity. Researching and writing articles like this takes a lot of time, so any and all tips are greatly appreciated!